
Chinese Journal OF Rice Science ›› 2020, Vol. 34 ›› Issue (2): 115-124.DOI: 10.16819/j.1001-7216.2020.9122
• Research Papers • Previous Articles Next Articles
Changjian WANG, Long CHEN, Liping DAI, Xueli LU, Jinli HE, Long YANG, Jiang HU, Li ZHU, Guojun DONG, Guangheng ZHANG, Zhenyu GAO, Deyong REN, Guang CHEN, Lan SHEN, Qiang ZHANG, Longbiao GUO, Qian QIAN*( ), Dali ZENG*(
), Dali ZENG*( )
)
Received:2019-11-15
Revised:2019-12-27
Online:2020-03-10
Published:2020-03-10
Contact:
Qian QIAN, Dali ZENG
About author:#These authors contributed equally to this work
王昌健, 陈龙, 代丽萍, 路雪丽, 贺金立, 杨龙, 胡江, 朱丽, 董国军, 张光恒, 高振宇, 任德勇, 陈光, 沈兰, 张强, 郭龙彪, 钱前*( ), 曾大力*(
), 曾大力*( )
)
通讯作者:
钱前,曾大力
作者简介:#共同第一作者
基金资助:CLC Number:
Changjian WANG, Long CHEN, Liping DAI, Xueli LU, Jinli HE, Long YANG, Jiang HU, Li ZHU, Guojun DONG, Guangheng ZHANG, Zhenyu GAO, Deyong REN, Guang CHEN, Lan SHEN, Qiang ZHANG, Longbiao GUO, Qian QIAN, Dali ZENG. Identification and Fine Mapping of Defective Pistil and Stamens 2 in Rice[J]. Chinese Journal OF Rice Science, 2020, 34(2): 115-124.
王昌健, 陈龙, 代丽萍, 路雪丽, 贺金立, 杨龙, 胡江, 朱丽, 董国军, 张光恒, 高振宇, 任德勇, 陈光, 沈兰, 张强, 郭龙彪, 钱前, 曾大力. 水稻花器官发育基因DPS2的鉴定和精细定位[J]. 中国水稻科学, 2020, 34(2): 115-124.
Add to citation manager EndNote|Ris|BibTeX
URL: http://www.ricesci.cn/EN/10.16819/j.1001-7216.2020.9122
| 引物名称 | 前引物 | 后引物 |
|---|---|---|
| Primer name | Forward primer(5'-3') | Reverse primer(5'-3') |
| BY-4 | GGAGGTGACTACGAAGGCATT | GAAGCTTATCCTTTTCACTTTCG |
| BY19 | GAACTCCAAGTCCACCCTGAT | GATAAATTCTGTTGGTGGGAGATTG |
| BY5 | GTTCACCAATAATCACCACAAGAGC | TAGCACCCCAGCAAGGACTCG |
| BY6-2 | CTCGGCTCTCTTTGCTCGG | TGCCGACGAGGTCACCGCC |
| BY7-3 | TATTTAGCCCTGGTTGAATGGT | TACCTAAGCTAAGTGGAAAGA |
| BY20 | GACCAGAGGTGGACCAAGGA | GTGAATGGAAAGTGTGTGAAAGATC |
| BY10-1 | GACTATATAGATATGTCCTCGGAAG | TTACCTATAACGAACCAAAGGC |
| BY13 | GGAAACAATATACTATGGTGTGGAT | CGTGGGTGGAGTTTAGAATG |
Table 1 Part of primers used in the research.
| 引物名称 | 前引物 | 后引物 |
|---|---|---|
| Primer name | Forward primer(5'-3') | Reverse primer(5'-3') |
| BY-4 | GGAGGTGACTACGAAGGCATT | GAAGCTTATCCTTTTCACTTTCG |
| BY19 | GAACTCCAAGTCCACCCTGAT | GATAAATTCTGTTGGTGGGAGATTG |
| BY5 | GTTCACCAATAATCACCACAAGAGC | TAGCACCCCAGCAAGGACTCG |
| BY6-2 | CTCGGCTCTCTTTGCTCGG | TGCCGACGAGGTCACCGCC |
| BY7-3 | TATTTAGCCCTGGTTGAATGGT | TACCTAAGCTAAGTGGAAAGA |
| BY20 | GACCAGAGGTGGACCAAGGA | GTGAATGGAAAGTGTGTGAAAGATC |
| BY10-1 | GACTATATAGATATGTCCTCGGAAG | TTACCTATAACGAACCAAAGGC |
| BY13 | GGAAACAATATACTATGGTGTGGAT | CGTGGGTGGAGTTTAGAATG |
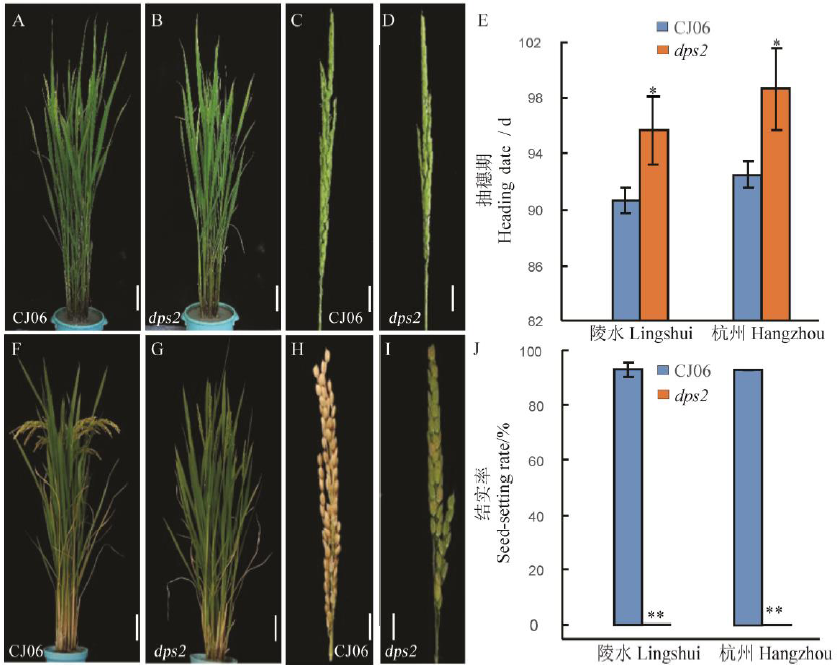
Fig. 1. Phenotypic comparison of dps2 mutant and wild type CJ06 at heading and maturity stage. A and B, Plants of Chunjiang 06(CJ06) and dps2 mutant at heading date; C and D, Panicle of CJ06 and dps2 at heading date; E, Heading date of CJ06 and dps2 in Lingshui, Hainan and Hangzhou, Zhejiang; F and G, Plants of CJ06 and dps2 at maturity stage; H and I, Panicle of CJ06 and dps2 at maturity stage; J, Seed-setting rate of CJ06 and dps2 in Lingshui and Hangzhou. Mean±SD(n=3). * and ** The difference between the wild type and mutant is significant at 0.05 and 0.01 level, respectively according to Student’s t test. In A, B, F, G, Bar=10 cm; In C, D, H, I, Bar=2 cm.

Fig. 2. Stamens phenotypic observation of wild type Chunjiang 06(CJ06) and dps2 mutant. A, Glumous flower of CJ06; B to D, Glumous flower of dps2 mutant; E, F, Anthers of the wild type and dps2; G, H, The outmost surface on epidermis anthers of the wild type and dps2; I, J, Pollens of CJ06 and dps2 mutant. Lo, Lodicule; An, Anther; F, Filament. From A to D, Bar=1 mm; In E, F, Bar=25 μm; In G, H, Bar=10 μm; In I, J, Bar=50 μm.
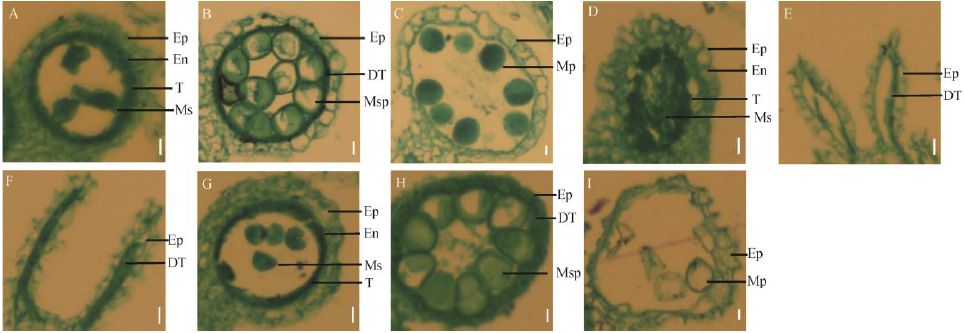
Fig. 3. Transverse section of anthers of dps2 mutant and wild type Chunjiang 06(CJ06) at different stages. A to C, CJ06; D to I, dps2 mutant; A, D, G, Cross-section of anther at the stage 9; B, E, H, Cross-section of anther at the stage 10; C, F, I, Cross-section of anther at the stage 12; Ep, Epidermis; En, Endothecium; T, Tapetum; DT, Degeneration tapetum; Ms, Microsporocyte; Msp, Microspore; Mp, Mature pollen. Bar=10 μm.
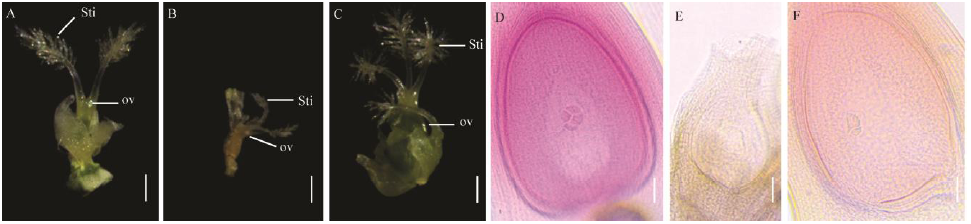
Fig. 4. Pistils phenotypic observation of wild type Chunjiang 06(CJ06) and dps2 mutant. A, Pistils of wild type; B, C, Pistils of dps2 mutant; D, Embryo sac of wild type; E, F, Embryo sac of dps2 mutant. Sti, Stigma; ov, Ovary. Scale bars: 1 mm in A, B and C, 50 μm in D to F.
| 杂交组合 Hybridized combination | F1 | F2 | χ2(3: 1) | χ2(0.05) | ||||
|---|---|---|---|---|---|---|---|---|
| 正常表型 Wild-type phenotype | 突变表型 dps2 phenotype | 正常表型 Wild-type phenotype | 突变表型 dps2 phenotype | |||||
| DPS2dps2/日本晴 DPS2dps2/Nipponbare | 7 | 0 | 236 | 79 | 0.001 | 3.84 | ||
| 日本晴/DPS2dps2 Nipponbare/DPS2dps2 | 4 | 0 | 133 | 43 | 0.030 | 3.84 | ||
| DPS2dps2/9311 | 5 | 0 | 160 | 42 | 1.908 | 3.84 | ||
| 9311/ DPS2dps2 | 4 | 0 | 127 | 35 | 0.996 | 3.84 | ||
Table 2 Genetic analysis of the dps2 locus
| 杂交组合 Hybridized combination | F1 | F2 | χ2(3: 1) | χ2(0.05) | ||||
|---|---|---|---|---|---|---|---|---|
| 正常表型 Wild-type phenotype | 突变表型 dps2 phenotype | 正常表型 Wild-type phenotype | 突变表型 dps2 phenotype | |||||
| DPS2dps2/日本晴 DPS2dps2/Nipponbare | 7 | 0 | 236 | 79 | 0.001 | 3.84 | ||
| 日本晴/DPS2dps2 Nipponbare/DPS2dps2 | 4 | 0 | 133 | 43 | 0.030 | 3.84 | ||
| DPS2dps2/9311 | 5 | 0 | 160 | 42 | 1.908 | 3.84 | ||
| 9311/ DPS2dps2 | 4 | 0 | 127 | 35 | 0.996 | 3.84 | ||
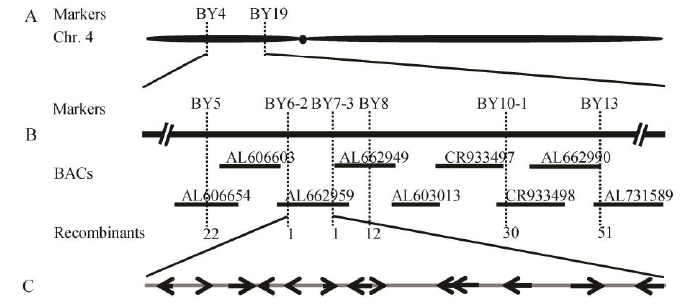
Fig. 5. Location of DPS2 on rice chromosome 4. A, Preliminary localization of DPS2 gene; B, Fine mapping of DPS2 gene; C, ORFs between markers BY6-2 and BY7-3.
| 登录号 | 位置 | 功能注释 |
|---|---|---|
| Gene accession | Location/bp | Functional annotation |
| LOC_Os04g08290 | 4 427 683-4 424 830 | ZOS4-04-C2H2锌指蛋白 ZOS4-04-C2H2 zinc finger protein |
| LOC_Os04g08300 | 4 433 624-4 433 839 | 反转录转座子蛋白,Ty3-gypsy亚类 Retrotransposon protein, putative, Ty3-gypsy subclass |
| LOC_Os04g08310 | 4 436 856-4 441 657 | 表达蛋白 Expressed protein |
| LOC_Os04g08320 | 4 444 004-4 441 881 | 假定蛋白YIF1B YIF1B, putative |
| LOC_Os04g08330 | 4 446 960-4 446 649 | 表达蛋白 Expressed protein |
| LOC_Os04g08340 | 4 452 696-4 455 350 | OsSigP3-假定的Ⅰ型信号肽酶同系物;采用假定的Ser/Lys催化二元体 OsSigP3-putative Type I signal peptidase homologue, employs a putative Ser/Lys catalytic dyad |
| LOC_Os04g08350 | 4 461 845-4 457 458 | 半胱氨酸合酶,叶绿体/染色质前体 Cysteine synthase, chloroplast/chromoplast precursor |
| LOC_Os04g08360 | 4 463 046-4 463 552 | 表达蛋白 Expressed protein |
| LOC_Os04g08370 | 4 473 512-4 470 642 | 富含亮氨酸的重复家族蛋白 Leucine rich repeat family protein |
| LOC_Os04g08390 | 4 490 101-4 484 997 | 富含亮氨酸的重复家族蛋白 Leucine rich repeat family protein |
| LOC_Os04g08400 | 4 497 789-4 503 741 | 反转录转座子蛋白 Retrotransposon protein |
| LOC_Os04g08410 | 4 513 883-4 508 804 | 含有ELM2结构域蛋白 ELM2 domain containing protein |
Table 3 The 12 genes in the candidate interval on chromosome 4.
| 登录号 | 位置 | 功能注释 |
|---|---|---|
| Gene accession | Location/bp | Functional annotation |
| LOC_Os04g08290 | 4 427 683-4 424 830 | ZOS4-04-C2H2锌指蛋白 ZOS4-04-C2H2 zinc finger protein |
| LOC_Os04g08300 | 4 433 624-4 433 839 | 反转录转座子蛋白,Ty3-gypsy亚类 Retrotransposon protein, putative, Ty3-gypsy subclass |
| LOC_Os04g08310 | 4 436 856-4 441 657 | 表达蛋白 Expressed protein |
| LOC_Os04g08320 | 4 444 004-4 441 881 | 假定蛋白YIF1B YIF1B, putative |
| LOC_Os04g08330 | 4 446 960-4 446 649 | 表达蛋白 Expressed protein |
| LOC_Os04g08340 | 4 452 696-4 455 350 | OsSigP3-假定的Ⅰ型信号肽酶同系物;采用假定的Ser/Lys催化二元体 OsSigP3-putative Type I signal peptidase homologue, employs a putative Ser/Lys catalytic dyad |
| LOC_Os04g08350 | 4 461 845-4 457 458 | 半胱氨酸合酶,叶绿体/染色质前体 Cysteine synthase, chloroplast/chromoplast precursor |
| LOC_Os04g08360 | 4 463 046-4 463 552 | 表达蛋白 Expressed protein |
| LOC_Os04g08370 | 4 473 512-4 470 642 | 富含亮氨酸的重复家族蛋白 Leucine rich repeat family protein |
| LOC_Os04g08390 | 4 490 101-4 484 997 | 富含亮氨酸的重复家族蛋白 Leucine rich repeat family protein |
| LOC_Os04g08400 | 4 497 789-4 503 741 | 反转录转座子蛋白 Retrotransposon protein |
| LOC_Os04g08410 | 4 513 883-4 508 804 | 含有ELM2结构域蛋白 ELM2 domain containing protein |
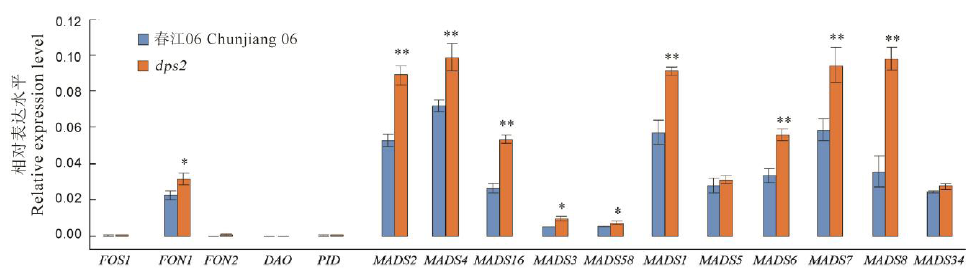
Fig. 6. Expression profile of the genes involved in the number and development of flower organs of dps2 mutant and wild type Chunjiang 06. Mean±SD(n=3). * and ** indicate the difference between the wild type Chunjiang 06 and mutant dps2 is significant at 0.05 and 0.01 levels, respectively according to Student’s t test.
| [1] | Pelaz S, Ditta G S, Baumann E, Wisman E, Yanofsky M F.B and C floral organ identity functions require SEPALLATA MADS-box genes[J]. Nature, 2000, 405(6783): 200-203. |
| [2] | Coen E S, Meyerowitz E M.The war of the whorls genetic interactions controlling flower development[J]. Nature, 1991, 353(6339): 31-37. |
| [3] | Ditta G, Pinyopich A, Robles P, Pelaz S, Yanofsky M F.The SEP4 gene of Arabidopsis thaliana functions in floral organ and meristem identity[J]. Current Biology, 2004, 14(21): 1935-1940. |
| [4] | Causier B, Schwarz-Sommer Z, Davies B.Floral organ identity: 20 years of ABCs[J]. Seminars in Cell and Developmental Biology, 2010, 21(1): 73-79. |
| [5] | Kalika P, Usha V.Double-stranded RNA interference of a rice PI/GLO paralog, OsMADS2, uncovers its second-whorl-specific function in floral organ patterning[J]. Genetics,2003, 165(4): 2301-2305. |
| [6] | Shri R Y, Kalika P, Usha V.Divergent regulatoryOsMADS2 functions control size, shape and differentiation of the highly derived rice floret second-whorl organ. Genetics, 2007, 176(1): 283. |
| [7] | Shan G Y, Shinnosuke O, Mayumi K, Hitoshi Y.Unequal genetic redundancy of rice PISTILLATA orthologs, OsMADS2 and OsMADS4, in lodicule and stamen development[J]. Plant & Cell Physiology, 2008, 49(5): 853. |
| [8] | Nagasawa M, Miyoshi M, Sano Y, Satoh H, Hirano H, Sakai H, Nagato Y.SUPERWOMAN1 and DROOPING LEAF genes control floral organ identify in rice[J]. Development, 2003, (130): 708-718 |
| [9] | Yun D P, Liang W Q, Dreni L D, Yin C S, Zhou Z G, Kater M M, Zhang D B.OsMADS16 genetically interacts with OsMADS3and OsMADS58 in specifying floral patterning in rice[J]. Molecular Plant, 2013, 6(3): 743-756. |
| [10] | Hu Y, Liang W Q, Yin C S, Yang X L, Ping B Z, Li A X, Jia R, Chen M J, Luo Z J, Cai Q, Zhao X X, Zhang D B, Yuan Z.Interactions of OsMADS1 with floral homeotic genes in rice flower development[J]. Molecular Plant, 2015, 8(9): 1366-1384. |
| [11] | Hu L F, Liang W Q, Yin C S, Cui X, Zong J, Wang X, Hu J P, Zhang D B.Rice MADS3 regulates ROS homeostasis during late anther development[J]. Plant Cell, 2011, 23(2): 515-533. |
| [12] | Yamaguchi T, Nagasawa N, Kawasaki S, Matsuoka M, Nagato Y, Hirano H Y.The YABBY gene DROOPING LEAF regulates carpel specification and midrib development in Oryza sativa[J]. Plant Cell, 2004, 16(2): 500-509. |
| [13] | Lopez-Dee ZP, Wittich P, Enrico P M, Rigola D, Del B I, Gorla M S, Kater M M, Colombo L.OsMADS13, a novel rice MADS-box gene expressed during ovule development[J]. Developmental Genetics, 2015, 25(3): 237-244. |
| [14] | Dreni L, Jacchia S, Fornara F, Fornari M, Ouwerkerk P B, An G, Colombo L, Kater M M.The D-lineage MADS-box gene OsMADS13 controls ovule identity in rice[J]. The Plant Journal, 2007, 52(4): 690-699. |
| [15] | Prasad K, Sriram P, Kumar C S, Kushalappa K, Vijayraghavan U.Ectopic expression of rice OsMADS1 reveals a role in specifying the lemma and palea, grass floral organs analogous to sepals[J]. Development Genes & Evolution, 2001, 211(6): 281-290. |
| [16] | Li H F, Liang W Q, Rui D J, Yin C S, Zong J, Kong H Z, Zhang D B.The AGL6-like gene OsMADS6 regulates floral organ and meristem identities in rice[J]. Cell Research, 2010, 20(3): 299. |
| [17] | Cui R F, Han J K, Zhao S Z, Su K M, Feng W, Du X Q, Xu Q J, Kang C, Theissen G, Zheng M.Functional conservation and diversification of class E floral homeotic genes in rice (Oryza sativa)[J]. Plant Journal, 2010, 61(5): 767-781. |
| [18] | Suzaki T, Sato M, Ashikari M, Miyoshi M, Nagato Y, Hirano H Y.The gene FLORAL ORGAN NUMBER1 regulates floral meristern size in rice and encodes a leucine-rich repeat receptor kinase orthologous to Arabidopsis CLAVATA1[J]. Development, 2004, 131(22): 5649-5657. |
| [19] | Suzaki T, Toriba T, Fujimoto M, Tsutsumi N, Kitano H, Hirano H Y.Conservation and diversification of meristem maintenance mechanism in Oryza sativa: Function of the FLORAL ORGAN NUMBER2 gene[J]. Plant & Cell Physiology, 2006, 47(12): 1591-1602. |
| [20] | Li J, Zhang W L, Xia Z H, Jiang G H, Qian Q, Li A L, Cheng Z K, Zhu L H, Long M, Zhai W X.A paracentric inversion suppresses genetic recombination at the FON3 locus with breakpoints corresponding to sequence gaps on rice chromosome 11L[J]. Molecular Genetics & Genomics ,2007, 277(3): 263-272. |
| [21] | 张向前, 邹金松, 朱海涛, 李晓燕, 曾瑞珍. 水稻早熟多子房突变体fon5的遗传分析和基因定位[J]. 遗传, 2008, 30(10): 1349-1355. |
| Zhang X Q, Zou J S, Zhu H T, Li X Y, Zeng R Z.Genetic analysis and gene mapping of an early flowering and multi-ovary mutant in rice (Oryza sativa L.)[J]. Hereditas, 2008, 30(10): 1349-1355. | |
| [22] | 赵福永,王洁雅, 黄显波, 邓则勤, 林成豹, 严寒, 田志宏. 水稻花器官数目突变体fon6的研究初报[J]. 杂交水稻, 2011, 26(2): 52-57. |
| Zhao F Y, Wang J Y, Huang X B, Deng Z Q, Lin C B, Han Y, Tian Z H.A preliminary study on the floral organ number mutant fon6 in rice[J]. Hybrid Rice, 2011, 26(2): 52-57. | |
| [23] | Thomson B, Zheng B, Wellmer F.Floral organogenesis: When knowing your ABCs is not enough[J]. Plant Physiol, 2017, 173(1): 56-64. |
| [24] | Livak K J, Schmittgen T D.Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method[J]. Methods, 2001, 25(4): 402-408. |
| [25] | Zhang D B, Wilson Z A.Stamen specification and anther development in rice[J]. Chinese Science Bulletin, 2009, 54(14): 2342-2353. |
| [26] | Xing Y Z, Zhang Q F.Genetic and molecular bases of rice yield[J]. Annual Review of Plant Biology, 2010, 61(1): 421-442. |
| [27] | Ikeda K, Sunohara H, Nagato Y.Development course of inflorescence and spikelet in rice[J]. Breeding Science, 2004, 54: 147-156. |
| [28] | 吴华茂. 水稻穗粒数相关QTL分析及水稻花器官发育突变体dps1的鉴定[D]. 中国科学院大学,2018. |
| Wu H M.Studies on rice QTL of grain number per panicle and mutant dps1 defective in floral organ development[D]. University of Chinese Academy of Science, 2018. | |
| [29] | Yamaguchi T, Lee D Y, Miyao A, Hirochika H, An G, Hirano H Y.Functional diversification of the two C-class MADS box genes OSMADS3 and OSMADS58 in Oryza sativa[J]. Plant Cell, 2006, 18(1): 15-28. |
| [30] | Moreau F, Thévenon E, Blanvillain R, Lopezvidriero I, Francozorrilla J M, Dumas R, Parcy F, Morel P, Trehin C, Carles C C.The Myb-domain protein ULTRAPETALA1 INTERACTING FACTOR 1 controls floral meristem activities in Arabidopsis[J]. Development, 2016, 143(7): 1108-1119. |
| [31] | Prunet N, Yang W B, Das P, Meyerowitz E M, Jack T P.SUPERMAN prevents class B gene expression and promotes stem cell termination in the fourth whorl of Arabidopsis thaliana flowers[J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114(27): 7166. |
| [32] | Sharma N, Xin R J, Kim D H, Sung S, Lange T, Huq E.NO FLOWERING IN SHORT DAY (NFL) is a bHLH transcription factor that promotes flowering specifically under short-day conditions in Arabidopsis[J]. Development, 2016, 143(4): 682. |
| [33] | Hugouvieux V, Silva C S, Jourdain A, Stigliani A, Charras Q, Conn V, Conn S J, Carles C C, Parcy F, Zubieta C.Tetramerization of MADS family transcription factors SEPALLATA3 and AGAMOUS is required for floral meristem determinacy in Arabidopsis[J]. Nucleic Acids Research, 2018. |
| [34] | Sommer R J, Retzlaff M, Goerlich K, Sander K, Tautz D.Evolutionary conservation pattern of zinc-finger domains of Drosophila segmentation genes[J]. Proceedings of the National Academy of Sciences of the United States of America, 1992, 89(22): 10782-10786. |
| [35] | Hiratsu K, Ohta M, Matsui K, Ohme-Takagi M.The SUPERMAN protein is an active repressor whose carboxy-terminal repression domain is required for the development of normal flowers[J]. Febs Letters, 2002, 514(2): 351-354. |
| [36] | Sakai H, Medrano LJ, Meyerowitz E M.Role of SUPERMAN in maintaining Arabidopsis floral whorl boundaries[J]. Nature, 1995, 378(6553): 199-203. |
| [37] | Gaiser J C, Robinson-Beers K, Gasser C S.The Arabidopsis SUPERMAN gene mediates asymmetric growth of the outer integument of ovules[J]. Plant Cell, 1995, 7(3): 333-345. |
| [38] | Nibau C, Stilio V S D, Wu H M, Cheung A Y. Arabidopsis and Tobacco SUPERMAN regulate hormone signalling and mediate cell proliferation and differentiation[J]. Journal of Experimental Botany, 2011, 62(3): 949-961. |
| [39] | Uemura A, Yamaguchi N, Xu Y, Wee W Y, Ichihashi Y, Suzuki T, Shibata A, Shirasu K, Ito T.Regulation of floral meristem activity through the interaction of AGAMOUS, SUPERMAN, and CLAVATA3 in Arabidopsis[J]. Plant Reproduction, 2017, 31(1): 89-105. |
| [1] | GUO Zhan, ZHANG Yunbo. Research Progress in Physiological,Biochemical Responses of Rice to Drought Stress and Its Molecular Regulation [J]. Chinese Journal OF Rice Science, 2024, 38(4): 335-349. |
| [2] | WEI Huanhe, MA Weiyi, ZUO Boyuan, WANG Lulu, ZHU Wang, GENG Xiaoyu, ZHANG Xiang, MENG Tianyao, CHEN Yinglong, GAO Pinglei, XU Ke, HUO Zhongyang, DAI Qigen. Research Progress in the Effect of Salinity, Drought, and Their Combined Stresses on Rice Yield and Quality Formation [J]. Chinese Journal OF Rice Science, 2024, 38(4): 350-363. |
| [3] | XU Danjie, LIN Qiaoxia, LI Zhengkang, ZHUANG Xiaoqian, LING Yu, LAI Meiling, CHEN Xiaoting, LU Guodong. OsOPR10 Positively Regulates Rice Blast and Bacterial Blight Resistance [J]. Chinese Journal OF Rice Science, 2024, 38(4): 364-374. |
| [4] | CHEN Mingliang, ZENG Xihua, SHEN Yumin, LUO Shiyou, HU Lanxiang, XIONG Wentao, XIONG Huanjin, WU Xiaoyan, XIAO Yeqing. Typing of Inter-subspecific Fertility Loci and Fertility Locus Pattern of indica-japonica Hybrid Rice [J]. Chinese Journal OF Rice Science, 2024, 38(4): 386-396. |
| [5] | DING Zhengquan, PAN Yueyun, SHI Yang, HUANG Haixiang. Comprehensive Evaluation and Comparative Analysis of Jiahe Series Long-Grain japonica Rice with High Eating Quality Based on Gene Chip Technology [J]. Chinese Journal OF Rice Science, 2024, 38(4): 397-408. |
| [6] | HOU Xiaoqin, WANG Ying, YU Bei, FU Weimeng, FENG Baohua, SHEN Yichao, XIE Hangjun, WANG Huanran, XU Yongqiang, WU Zhihai, WANG Jianjun, TAO Longxing, FU Guanfu. Mechanisms Behind the Role of Potassium Fulvic Acid in Enhancing Salt Tolerance in Rice Seedlings [J]. Chinese Journal OF Rice Science, 2024, 38(4): 409-421. |
| [7] | LÜ Zhou, YI Binghuai, CHEN Pingping, ZHOU Wenxin, TANG Wenbang, YI Zhenxie. Effects of Nitrogen Application Rate and Transplanting Density on Yield Formation of Small Seed Hybrid Rice [J]. Chinese Journal OF Rice Science, 2024, 38(4): 422-436. |
| [8] | HU Jijie, HU Zhihua, ZHANG Junhua, CAO Xiaochuang, JIN Qianyu, ZHANG Zhiyuan, ZHU Lianfeng. Effects of Rhizosphere Saturated Dissolved Oxygen on Photosynthetic and Growth Characteristics of Rice at Tillering Stage [J]. Chinese Journal OF Rice Science, 2024, 38(4): 437-446. |
| [9] | WU Yue, LIANG Chengwei, ZHAO Chenfei, SUN Jian, MA Dianrong. Occurrence of Weedy Rice Disaster and Ecotype Evolution in Direct-Seeded Rice Fields [J]. Chinese Journal OF Rice Science, 2024, 38(4): 447-455. |
| [10] | LIU Fuxiang, ZHEN Haoyang, PENG Huan, ZHENG Liuchun, PENG Deliang, WEN Yanhua. Investigation and Species Identification of Cyst Nematode Disease on Rice in Guangdong Province [J]. Chinese Journal OF Rice Science, 2024, 38(4): 456-461. |
| [11] | CHEN Haotian, QIN Yuan, ZHONG Xiaohan, LIN Chenyu, QIN Jinghang, YANG Jianchang, ZHANG Weiyang. Research Progress on the Relationship Between Rice Root, Soil Properties and Methane Emissions in Paddy Fields [J]. Chinese Journal OF Rice Science, 2024, 38(3): 233-245. |
| [12] | MIAO Jun, RAN Jinhui, XU Mengbin, BO Liubing, WANG Ping, LIANG Guohua, ZHOU Yong. Overexpression of RGG2, a Heterotrimeric G Protein γ Subunit-Encoding Gene, Improves Drought Tolerance in Rice [J]. Chinese Journal OF Rice Science, 2024, 38(3): 246-255. |
| [13] | YIN Xiaoxiao, ZHANG Zhihan, YAN Xiulian, LIAO Rong, YANG Sijia, Beenish HASSAN, GUO Daiming, FAN Jing, ZHAO Zhixue, WANG Wenming. Signal Peptide Validation and Expression Analysis of Multiple Effectors from Ustilaginoidea virens [J]. Chinese Journal OF Rice Science, 2024, 38(3): 256-265. |
| [14] | ZHU Yujing, GUI Jinxin, GONG Chengyun, LUO Xinyang, SHI Jubin, ZHANG Haiqing, HE Jiwai. QTL Mapping for Tiller Angle in Rice by Genome-wide Association Analysis [J]. Chinese Journal OF Rice Science, 2024, 38(3): 266-276. |
| [15] | WEI Qianqian, WANG Yulei, KONG Haimin, XU Qingshan, YAN Yulian, PAN Lin, CHI Chunxin, KONG Yali, TIAN Wenhao, ZHU Lianfeng, CAO Xiaochuang, ZHANG Junhua, ZHU Chunqun. Mechanism of Hydrogen Sulfide, a Signaling Molecule Involved in Reducing the Inhibitory Effect of Aluminum Toxicity on Rice Growth Together with Sulfur Fertilizer [J]. Chinese Journal OF Rice Science, 2024, 38(3): 290-302. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||