
中国水稻科学 ›› 2025, Vol. 39 ›› Issue (5): 624-634.DOI: 10.16819/j.1001-7216.2025.240106
韶也1,2,#, 胡远艺1,2,#, 彭彦1,2,#, 毛毕刚1,2, 刘慧敏1,2, 唐婵娟1, 雷斌1,2, 唐丽1,2, 余丽霞3, 李文建3, 罗武中3, 罗治斌1,2, 袁远涛1,2, 李曜魁1,2, 张丹1,2, 周利斌3, 柏连阳2,4,*( ), 唐文帮1,2,*(
), 唐文帮1,2,*( ), 赵炳然1,2,*(
), 赵炳然1,2,*( )
)
收稿日期:2024-01-09
修回日期:2024-04-17
出版日期:2025-09-10
发布日期:2025-09-10
通讯作者:
*email:lybai@hunaas.cn,email:tangwenbang@163.com,email:brzhao652@hhrrc.ac.cn
作者简介:#共同第一作者
基金资助:
SHAO Ye1,2,#, HU Yuanyi1,2,#, PENG Yan1,2,#, MAO Bigang1,2, LIU Huimin1,2, TANG Chanjuan1, LEI Bin1,2, TANG Li1,2, YU Lixia3, LI Wenjian3, LUO Wuzhong3, LUO Zhibin1,2, YUAN Yuantao1,2, LI Yaokui1,2, ZHANG Dan1,2, ZHOU Libin3, BAI Lianyang2,4,*( ), TANG Wenbang1,2,*(
), TANG Wenbang1,2,*( ), ZHAO Bingran1,2,*(
), ZHAO Bingran1,2,*( )
)
Received:2024-01-09
Revised:2024-04-17
Online:2025-09-10
Published:2025-09-10
Contact:
*email:lybai@hunaas.cn,email:tangwenbang@163.com,email:brzhao652@hhrrc.ac.cn
About author:#These authors contributed equally to this work
摘要:
【目的】以两系杂交水稻卓两优1126为“底盘品种”,快速创制具有镉低积累、香味、耐储藏、耐淹、低升糖指数等性状的改良品种。【方法】12C6+碳离子束诱变卓两优1126亲本卓234S和湘农恢1126,利用M1TDS技术从诱变M1群体中鉴定OsNRAMP5、OsBADH2、OsLOX3、OsPAO5、OsSSIIIa和OsBEIIb基因嵌合突变株,通过KASP分型从诱变M2群体中鉴定分离目标基因纯合(杂合)突变株,利用OsNRAMP5突变亲本配制OsNRAMP5突变组合。【结果】从重离子束诱变M1群体中鉴定到13株为5个目标基因突变的嵌合突变株,其中7个M2株系中鉴定到目标基因的纯合或杂合突变;OsNRAMP5纯合突变株镉含量较原始亲本显著降低,OsBADH2纯合突变株香味特征化合物2-乙酰基吡咯啉(2-AP)含量较原始亲本显著上升;OsNRAMP5基因突变亲本配制的组合,在镉污染土(土壤有效镉含量0.677 mg/kg,pH 5.6)盆栽种植,其籽粒镉含量为0.05 mg/kg,而原始组合为0.91 mg/kg。【结论】利用“重离子束诱变+M1TDS技术”实现了卓两优1126镉低积累等性状的快速改良,为生物传统诱变育种进化到定向诱变育种提拱了成功案例和共性技术参考。
韶也, 胡远艺, 彭彦, 毛毕刚, 刘慧敏, 唐婵娟, 雷斌, 唐丽, 余丽霞, 李文建, 罗武中, 罗治斌, 袁远涛, 李曜魁, 张丹, 周利斌, 柏连阳, 唐文帮, 赵炳然. 基于M1TDS靶向筛选技术的重离子束诱变定向改良杂交水稻卓两优1126性状的研究[J]. 中国水稻科学, 2025, 39(5): 624-634.
SHAO Ye, HU Yuanyi, PENG Yan, MAO Bigang, LIU Huimin, TANG Chanjuan, LEI Bin, TANG Li, YU Lixia, LI Wenjian, LUO Wuzhong, LUO Zhibin, YUAN Yuantao, LI Yaokui, ZHANG Dan, ZHOU Libin, BAI Lianyang, TANG Wenbang, ZHAO Bingran. Directed Improvement of Hybrid Rice Zhuoliangyou 1126 by Heavy Ion Beam Mutagenesis Based on M1TDS Targeted Screening Technology[J]. Chinese Journal OF Rice Science, 2025, 39(5): 624-634.
| 名称 Name | 序列 Sequence |
|---|---|
| OsNRAMP5-246-8-F | GAAGGTGACCAAGTTCATGCTTGTGGTTGGCTCCGGCTTAA |
| OsNRAMP5-246-8-H | GAAGGTCGGAGTCAACGGATTTGTGGTTGGCTCCGGCTTGC |
| OsNRAMP5-246-8-C | CACAAAATGAAACAGTGGAAACCGATC |
| OsNRAMP5-234-6-F | GAAGGTGACCAAGTTCATGCTCACAAAATGAAACAGTGGAAACCGAT |
| OsNRAMP5-234-6-H | GAAGGTCGGAGTCAACGGATTCACAAAATGAAACAGTGGAAACCGAC |
| OsNRAMP5-234-6-C | CGTACCTCATATCTGTGGTTGGCTC |
| OsNRAMP5-204-6-F | GAAGGTGACCAAGTTCATGCTCACAAAATGAAACAGTGGAAACCGA |
| OsNRAMP5-204-6-H | GAAGGTCGGAGTCAACGGATTCACAAAATGAAACAGTGGAAACCGC |
| OsNRAMP5-204-6-C | CGTACCTCATATCTGTGGTTGGCTC |
| OsNRAMP5-37-7-F | GAAGGTGACCAAGTTCATGCTTGAAGAAGCTAAGAGAGGAAGCAACAA |
| OsNRAMP5-37-7-H | GAAGGTCGGAGTCAACGGATTTGAAGAAGCTAAGAGAGGAAGCAACAG |
| OsNRAMP5-37-7-C | ACTAGCTCTCCAGCTGATGCTC |
| OsBADH2-168-1-F | GAAGGTGACCAAGTTCATGCTGTGTAGTTGGGTTGATCACACCTT |
| OsBADH2-168-1-H | GAAGGTCGGAGTCAACGGATTGTGTAGTTGGGTTGATCACACCTC |
| OsBADH2-168-1-C | TGGGTCATAAATAAATATAAGCGCAGG |
| OsBADH2-224-8-F | GAAGGTGACCAAGTTCATGCTTTCGTCTTTTCTTGACAGCCTGTT |
| OsBADH2-224-8-H | GAAGGTCGGAGTCAACGGATTTTCGTCTTTTCTTGACAGCCTGTC |
| OsBADH2-224-8-C | AGGACTTTTTCCACCAAGTTCCAG |
| OsLOX3-760-7-F | GAAGGTGACCAAGTTCATGCTTATCTTCACCTATGCCACAAGGC |
| OsLOX3-760-7-H | GAAGGTCGGAGTCAACGGATTTATCTTCACCTATGCCACAAGGA |
| OsLOX3-760-7-C | CAGGGTATCATCATCACGTAAGAAC |
| OsLOX3-115-5-F | GAAGGTGACCAAGTTCATGCTGAGAGCAAAGTGTTGAACATGAAC |
| OsLOX3-115-5-H | GAAGGTCGGAGTCAACGGATTGAGAGCAAAGTGTTGAACATGAAG |
| OsLOX3-115-5-C | GGACCGACCCGGTTCTTGAG |
| OsPAO5-549-5-F | GAAGGTGACCAAGTTCATGCTTGCTTGACAGGAATCCACACCTA |
| OsPAO5-549-5-H | GAAGGTCGGAGTCAACGGATTTGCTTGACAGGAATCCACACCTG |
| OsPAO5-549-5-C | GCACCACCATTCCAGATATAGGTG |
| OsPAO5-1028-4-F | GAAGGTGACCAAGTTCATGCTAACTTTCCTCCAGGGTTACCAAAAT |
| OsPAO5-1028-4-H | GAAGGTCGGAGTCAACGGATTAACTTTCCTCCAGGGTTACCAAATC |
| OsPAO5-1028-4-C | GCTTGTCCCATCTTCAACACATAC |
| OsPAO5-84-2-F | GAAGGTGACCAAGTTCATGCTCTCAGCTCAAGAAAATGCTACCAGG |
| OsPAO5-84-2-H | GAAGGTCGGAGTCAACGGATTCTCAGCTCAAGAAAATGCTACCACT |
| OsPAO5-84-2-C | GACAGTACTCAACTGTACATGGTCTG |
| OsSSIIIa-598-7-F | GAAGGTGACCAAGTTCATGCTCCATTTGGTTCAAGGCCTAGAACTC |
| OsSSIIIa-598-7-H | GAAGGTCGGAGTCAACGGATTCCATTTGGTTCAAGGCCTAGAACTG |
| OsSSIIIa-598-7-C | CACTGTTTTCGACGTAGACCATG |
| OsBEIIb-931-5-F | GAAGGTGACCAAGTTCATGCTGTGTGATTGTTATTAAATCTCACCAGGA |
| OsBEIIb-931-5-H | GAAGGTCGGAGTCAACGGATTGTGTGATTGTTATTAAATCTCACCAGGC |
| OsBEIIb-931-5-C | GGCTATCTTAACTTTATGGGAAATGAGTTC |
表1 KASP基因分型引物序列
Table 1. Primer sequence for KASP genotyping
| 名称 Name | 序列 Sequence |
|---|---|
| OsNRAMP5-246-8-F | GAAGGTGACCAAGTTCATGCTTGTGGTTGGCTCCGGCTTAA |
| OsNRAMP5-246-8-H | GAAGGTCGGAGTCAACGGATTTGTGGTTGGCTCCGGCTTGC |
| OsNRAMP5-246-8-C | CACAAAATGAAACAGTGGAAACCGATC |
| OsNRAMP5-234-6-F | GAAGGTGACCAAGTTCATGCTCACAAAATGAAACAGTGGAAACCGAT |
| OsNRAMP5-234-6-H | GAAGGTCGGAGTCAACGGATTCACAAAATGAAACAGTGGAAACCGAC |
| OsNRAMP5-234-6-C | CGTACCTCATATCTGTGGTTGGCTC |
| OsNRAMP5-204-6-F | GAAGGTGACCAAGTTCATGCTCACAAAATGAAACAGTGGAAACCGA |
| OsNRAMP5-204-6-H | GAAGGTCGGAGTCAACGGATTCACAAAATGAAACAGTGGAAACCGC |
| OsNRAMP5-204-6-C | CGTACCTCATATCTGTGGTTGGCTC |
| OsNRAMP5-37-7-F | GAAGGTGACCAAGTTCATGCTTGAAGAAGCTAAGAGAGGAAGCAACAA |
| OsNRAMP5-37-7-H | GAAGGTCGGAGTCAACGGATTTGAAGAAGCTAAGAGAGGAAGCAACAG |
| OsNRAMP5-37-7-C | ACTAGCTCTCCAGCTGATGCTC |
| OsBADH2-168-1-F | GAAGGTGACCAAGTTCATGCTGTGTAGTTGGGTTGATCACACCTT |
| OsBADH2-168-1-H | GAAGGTCGGAGTCAACGGATTGTGTAGTTGGGTTGATCACACCTC |
| OsBADH2-168-1-C | TGGGTCATAAATAAATATAAGCGCAGG |
| OsBADH2-224-8-F | GAAGGTGACCAAGTTCATGCTTTCGTCTTTTCTTGACAGCCTGTT |
| OsBADH2-224-8-H | GAAGGTCGGAGTCAACGGATTTTCGTCTTTTCTTGACAGCCTGTC |
| OsBADH2-224-8-C | AGGACTTTTTCCACCAAGTTCCAG |
| OsLOX3-760-7-F | GAAGGTGACCAAGTTCATGCTTATCTTCACCTATGCCACAAGGC |
| OsLOX3-760-7-H | GAAGGTCGGAGTCAACGGATTTATCTTCACCTATGCCACAAGGA |
| OsLOX3-760-7-C | CAGGGTATCATCATCACGTAAGAAC |
| OsLOX3-115-5-F | GAAGGTGACCAAGTTCATGCTGAGAGCAAAGTGTTGAACATGAAC |
| OsLOX3-115-5-H | GAAGGTCGGAGTCAACGGATTGAGAGCAAAGTGTTGAACATGAAG |
| OsLOX3-115-5-C | GGACCGACCCGGTTCTTGAG |
| OsPAO5-549-5-F | GAAGGTGACCAAGTTCATGCTTGCTTGACAGGAATCCACACCTA |
| OsPAO5-549-5-H | GAAGGTCGGAGTCAACGGATTTGCTTGACAGGAATCCACACCTG |
| OsPAO5-549-5-C | GCACCACCATTCCAGATATAGGTG |
| OsPAO5-1028-4-F | GAAGGTGACCAAGTTCATGCTAACTTTCCTCCAGGGTTACCAAAAT |
| OsPAO5-1028-4-H | GAAGGTCGGAGTCAACGGATTAACTTTCCTCCAGGGTTACCAAATC |
| OsPAO5-1028-4-C | GCTTGTCCCATCTTCAACACATAC |
| OsPAO5-84-2-F | GAAGGTGACCAAGTTCATGCTCTCAGCTCAAGAAAATGCTACCAGG |
| OsPAO5-84-2-H | GAAGGTCGGAGTCAACGGATTCTCAGCTCAAGAAAATGCTACCACT |
| OsPAO5-84-2-C | GACAGTACTCAACTGTACATGGTCTG |
| OsSSIIIa-598-7-F | GAAGGTGACCAAGTTCATGCTCCATTTGGTTCAAGGCCTAGAACTC |
| OsSSIIIa-598-7-H | GAAGGTCGGAGTCAACGGATTCCATTTGGTTCAAGGCCTAGAACTG |
| OsSSIIIa-598-7-C | CACTGTTTTCGACGTAGACCATG |
| OsBEIIb-931-5-F | GAAGGTGACCAAGTTCATGCTGTGTGATTGTTATTAAATCTCACCAGGA |
| OsBEIIb-931-5-H | GAAGGTCGGAGTCAACGGATTGTGTGATTGTTATTAAATCTCACCAGGC |
| OsBEIIb-931-5-C | GGCTATCTTAACTTTATGGGAAATGAGTTC |
| 名称 Name | 序列 Sequence |
|---|---|
| OsNRAMP5-3E-540F | GTTGGTCCTGGATTCATGGTGTC |
| OsNRAMP5-3E-540R | TCCAATCAGAATCACCCAGAGCAG |
| OsNRAMP5-1E-265F | TCTTCGTCTACTTCCAGCTAGCC |
| OsNRAMP5-1E-265R | ACAGTGAACAACTGTCCATGGTG |
| OsBADH-4-471F | TTGATGAAGCAGCATGGGACATG |
| OsBADH-4-471R | GCTGCTAGGTACAATTTGTGAGAC |
| OsBADH-8-357F | CTTCAGCTGCTCCTATGGTTAAGG |
| OsBADH-8-357R | TCTAGCATCCAGCTCAGTTAAGTGC |
| OsLOX3-8-535F | GAGAAGAACCTCGAAGGCCTCAG |
| OsLOX3-8-535R | CCATCACAGCATGTGTGTTGAGC |
| OsPAO-6/7-563F | GCTCTCTTTGATAAGGATGGTCGTC |
| OsPAO-6/7-563R | ATGGCCACCAGTAAGAACATGTTC |
| OsBEIIb-17-359F | GGCATGAGAACCTCACATACTG |
| OsBEIIb-17-359R | GGAAGTACTTGTGGAGCTCTTGG |
表2 Sanger测序引物序列
Table 2. Primers for Sanger sequencing
| 名称 Name | 序列 Sequence |
|---|---|
| OsNRAMP5-3E-540F | GTTGGTCCTGGATTCATGGTGTC |
| OsNRAMP5-3E-540R | TCCAATCAGAATCACCCAGAGCAG |
| OsNRAMP5-1E-265F | TCTTCGTCTACTTCCAGCTAGCC |
| OsNRAMP5-1E-265R | ACAGTGAACAACTGTCCATGGTG |
| OsBADH-4-471F | TTGATGAAGCAGCATGGGACATG |
| OsBADH-4-471R | GCTGCTAGGTACAATTTGTGAGAC |
| OsBADH-8-357F | CTTCAGCTGCTCCTATGGTTAAGG |
| OsBADH-8-357R | TCTAGCATCCAGCTCAGTTAAGTGC |
| OsLOX3-8-535F | GAGAAGAACCTCGAAGGCCTCAG |
| OsLOX3-8-535R | CCATCACAGCATGTGTGTTGAGC |
| OsPAO-6/7-563F | GCTCTCTTTGATAAGGATGGTCGTC |
| OsPAO-6/7-563R | ATGGCCACCAGTAAGAACATGTTC |
| OsBEIIb-17-359F | GGCATGAGAACCTCACATACTG |
| OsBEIIb-17-359R | GGAAGTACTTGTGGAGCTCTTGG |
| 目标基因 Target gene | 基因编号 Gene ID | 目标表型 Target phenotype | 外显子数 No. of exons | 目标区域覆盖度 Coverage of target area(%) | 平均测序深度 Average sequencing depth |
|---|---|---|---|---|---|
| OsNRAMP5 | Os07g0257200 | 镉低积累 Low cadmium accumulation | 13 | 100 | 53398× |
| OsBADH2 | Os08g0424500 | 香味Aroma | 15 | 100 | 53086× |
| OsLOX3 | Os03g0699700 | 耐储藏Storage-tolerance | 9 | 100 | 50202× |
| OsPAO5 | Os04g0671300 | 耐淹 Submergence-tolerance | 10 | 100 | 54263× |
| OsSSIIIa | Os08g0191433 | 低升糖指数 Low glycemic index (GI) | 14 | 100 | 54510× |
| OsBEIIb | Os02g0528200 | 低升糖指数 Low glycemic index (GI) | 22 | 100 | 52192× |
表3 目标基因及靶向测序分析情况
Table 3. Target gene and targeted sequencing analysis
| 目标基因 Target gene | 基因编号 Gene ID | 目标表型 Target phenotype | 外显子数 No. of exons | 目标区域覆盖度 Coverage of target area(%) | 平均测序深度 Average sequencing depth |
|---|---|---|---|---|---|
| OsNRAMP5 | Os07g0257200 | 镉低积累 Low cadmium accumulation | 13 | 100 | 53398× |
| OsBADH2 | Os08g0424500 | 香味Aroma | 15 | 100 | 53086× |
| OsLOX3 | Os03g0699700 | 耐储藏Storage-tolerance | 9 | 100 | 50202× |
| OsPAO5 | Os04g0671300 | 耐淹 Submergence-tolerance | 10 | 100 | 54263× |
| OsSSIIIa | Os08g0191433 | 低升糖指数 Low glycemic index (GI) | 14 | 100 | 54510× |
| OsBEIIb | Os02g0528200 | 低升糖指数 Low glycemic index (GI) | 22 | 100 | 52192× |
| 目标基因 Target gene | 亲本 Parent | 混池编号 No. of the pool | 突变位置 Mutation site | 野生/突变基因型 Wild/mutant genotype |
|---|---|---|---|---|
| OsNRAMP5 | 卓234S Zhuo 234S | 217-288E | 第3外显子Exon 3 | C/CTTAA |
| 卓234S Zhuo 234S | 217-288W | 第3外显子Exon 3 | C/CTTAA | |
| 卓234S Zhuo 234S | 217-288E | 第3外显子Exon 3 | GA/G | |
| 湘农恢1126 Xiangnonghui 1126 | 145-216S | 第3外显子Exon 3 | GCAGAT/G | |
| 湘农恢1126 Xiangnonghui 1126 | 1-72N | 第1外显子Exon 1 | CTCAATCTCCAT/C | |
| OsBADH2 | 卓234S Zhuo 234S | 145-216E | 第4外显子和第4内含子Exon 4/Intron 4 | CCTTGGTATTTCACATTTTTCT/C |
| 湘农恢1126 Xiangnonghui 1126 | 217-238S | 第8外显子Exon 8 | GTT/G | |
| OsLOX3 | 卓234S Zhuo 234S | 718-789W | 第7外显子Exon 7 | G/GC |
| 湘农恢1126 Xiangnonghui 1126 | 73-144S | 第9外显子Exon 9 | GAAC/G | |
| 湘农恢1126 Xiangnonghui 1126 | 73-144E | 第9外显子Exon 9 | GAAC/G | |
| OsPAO5 | 卓234S Zhuo 234S | 504-575W | 第6外显子和第6内含子Exon 6/Intron 6 | CACTTACTTT/C |
| 卓234S Zhuo 234S | 1004-1040E | 第9外显子Exon 9 | ATGAT/A | |
| 卓234S Zhuo 234S | 991-1040W | 第9外显子Exon 9 | ATGAT/A | |
| 湘农恢1126 Xiangnonghui 1126 | 73-144E | 第9外显子Exon 9 | GTAGCTCC/G | |
| OsSSIIIa | 卓234S Zhuo 234S | 576-647E | 第13外显子Exon 13 | CTCG/C |
| OsBEIIb | 卓234S Zhuo 234S | 863-932W | 第17外显子Exon 17 | GATGT/G |
表4 混池目标基因靶向测序捕获突变情况
Table 4. Mutations were captured by mixed-pool targeted gene sequencing
| 目标基因 Target gene | 亲本 Parent | 混池编号 No. of the pool | 突变位置 Mutation site | 野生/突变基因型 Wild/mutant genotype |
|---|---|---|---|---|
| OsNRAMP5 | 卓234S Zhuo 234S | 217-288E | 第3外显子Exon 3 | C/CTTAA |
| 卓234S Zhuo 234S | 217-288W | 第3外显子Exon 3 | C/CTTAA | |
| 卓234S Zhuo 234S | 217-288E | 第3外显子Exon 3 | GA/G | |
| 湘农恢1126 Xiangnonghui 1126 | 145-216S | 第3外显子Exon 3 | GCAGAT/G | |
| 湘农恢1126 Xiangnonghui 1126 | 1-72N | 第1外显子Exon 1 | CTCAATCTCCAT/C | |
| OsBADH2 | 卓234S Zhuo 234S | 145-216E | 第4外显子和第4内含子Exon 4/Intron 4 | CCTTGGTATTTCACATTTTTCT/C |
| 湘农恢1126 Xiangnonghui 1126 | 217-238S | 第8外显子Exon 8 | GTT/G | |
| OsLOX3 | 卓234S Zhuo 234S | 718-789W | 第7外显子Exon 7 | G/GC |
| 湘农恢1126 Xiangnonghui 1126 | 73-144S | 第9外显子Exon 9 | GAAC/G | |
| 湘农恢1126 Xiangnonghui 1126 | 73-144E | 第9外显子Exon 9 | GAAC/G | |
| OsPAO5 | 卓234S Zhuo 234S | 504-575W | 第6外显子和第6内含子Exon 6/Intron 6 | CACTTACTTT/C |
| 卓234S Zhuo 234S | 1004-1040E | 第9外显子Exon 9 | ATGAT/A | |
| 卓234S Zhuo 234S | 991-1040W | 第9外显子Exon 9 | ATGAT/A | |
| 湘农恢1126 Xiangnonghui 1126 | 73-144E | 第9外显子Exon 9 | GTAGCTCC/G | |
| OsSSIIIa | 卓234S Zhuo 234S | 576-647E | 第13外显子Exon 13 | CTCG/C |
| OsBEIIb | 卓234S Zhuo 234S | 863-932W | 第17外显子Exon 17 | GATGT/G |
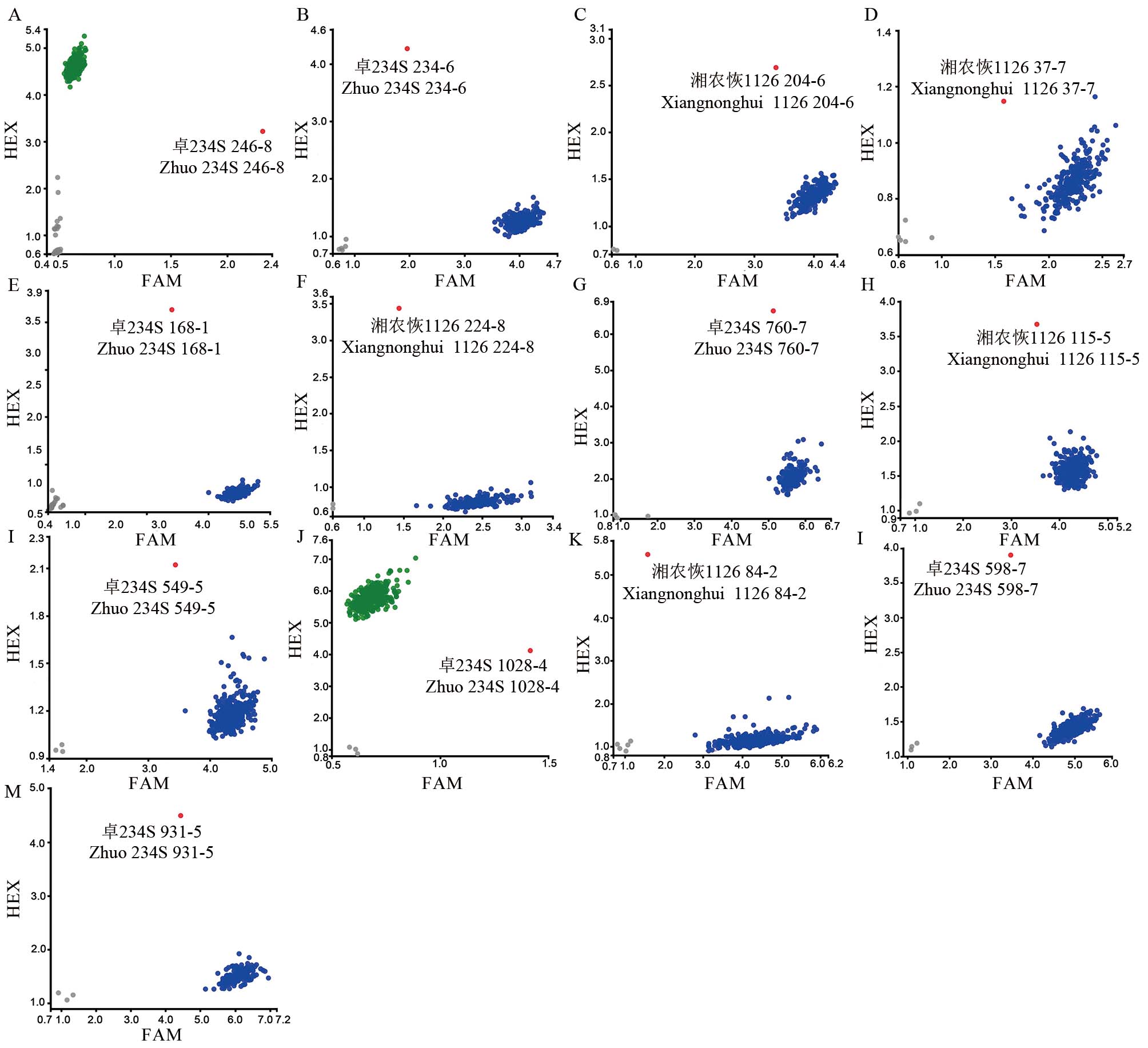
图1 目标基因突变混池的KASP基因分型 A~D分别代表OsNRAMP5基因不同突变位点基因分型;E, F 分别代表OsBADH2基因不同突变位点基因分型;G, H分别代表OsLOX3基因不同突变位点基因分型;I~K分别代表OsPAO5基因不同突变位点基因分型;L, M分别代表OsSSIIIa基因和OsBEIIb基因突变位点基因分型;A. 图中246-8代表诱变M1群体246行第8株(下同);蓝点/绿点代表野生基因型,红点代表突变基因型,灰点代表阴性对照;FAM和HEX分别代表两种不同颜色的荧光修饰标签。
Fig. 1. KASP genotyping in the mixed pool of target gene mutations A-D represent the genotyping of different mutation sites of OsNRAMP5 gene; E and F represent the genotyping of different mutation sites of OsBADH2 gene, respectively; G and H represent genotyping of different mutation sites of OsLOX3 gene, respectively; I-K represent genotyping of different mutation sites of OsPAO5 gene; L, M represent the genotyping of mutation sites in OsSSIIIa gene and OsBEIIb gene, respectively; In panel A, 246-8 represents the 8th plant in 246 rows of M1 population (the same below); Blue/green dots represent wild genotypes, red dots represent mutant genotypes, and gray dots represent negative controls; FAM and HEX represent fluorescently modified labels of two different colors, respectively.
| 基因 Gene | 品种 Variety | 突变株定位(行-株) Location of mutants (row-plant) | 突变分蘖占比 Proportion of tillers containing mutations |
|---|---|---|---|
| OsNRAMP5 | 卓234S Zhuo 234S | 246-8 | 18/18 |
| OsNRAMP5 | 卓234S Zhuo 234S | 234-6 | 4/18 |
| OsNRAMP5 | 湘农恢1126 Xiangnonghui 1126 | 204-6 | 3/6 |
| OsNRAMP5 | 湘农恢1126 Xiangnonghui 1126 | 37-7 | 1/8 |
| OsBADH2 | 卓234S Zhuo 234S | 168-1 | 3/18 |
| OsBADH2 | 湘农恢1126 Xiangnonghui 1126 | 224-8 | 1/6 |
| OsLOX3 | 卓234S Zhuo 234S | 760-7 | 1/18 |
| OsLOX3 | 湘农恢1126 Xiangnonghui 1126 | 115-5 | 2/6 |
| OsPAO5 | 卓234S Zhuo 234S | 549-5 | 1/18 |
| OsPAO5 | 卓234S Zhuo 234S | 1028-4 | 5/18 |
| OsPAO5 | 湘农恢1126 Xiangnonghui 1126 | 84-2 | 1/6 |
| OsSSIIIa | 卓234S Zhuo 234S | 598-7 | 1/18 |
| OsBEIIb | 卓234S Zhuo 234S | 931-5 | 4/18 |
表5 诱变M1突变株分蘖比例
Table 5. Tiller ratio of mutagenic M1 mutants
| 基因 Gene | 品种 Variety | 突变株定位(行-株) Location of mutants (row-plant) | 突变分蘖占比 Proportion of tillers containing mutations |
|---|---|---|---|
| OsNRAMP5 | 卓234S Zhuo 234S | 246-8 | 18/18 |
| OsNRAMP5 | 卓234S Zhuo 234S | 234-6 | 4/18 |
| OsNRAMP5 | 湘农恢1126 Xiangnonghui 1126 | 204-6 | 3/6 |
| OsNRAMP5 | 湘农恢1126 Xiangnonghui 1126 | 37-7 | 1/8 |
| OsBADH2 | 卓234S Zhuo 234S | 168-1 | 3/18 |
| OsBADH2 | 湘农恢1126 Xiangnonghui 1126 | 224-8 | 1/6 |
| OsLOX3 | 卓234S Zhuo 234S | 760-7 | 1/18 |
| OsLOX3 | 湘农恢1126 Xiangnonghui 1126 | 115-5 | 2/6 |
| OsPAO5 | 卓234S Zhuo 234S | 549-5 | 1/18 |
| OsPAO5 | 卓234S Zhuo 234S | 1028-4 | 5/18 |
| OsPAO5 | 湘农恢1126 Xiangnonghui 1126 | 84-2 | 1/6 |
| OsSSIIIa | 卓234S Zhuo 234S | 598-7 | 1/18 |
| OsBEIIb | 卓234S Zhuo 234S | 931-5 | 4/18 |
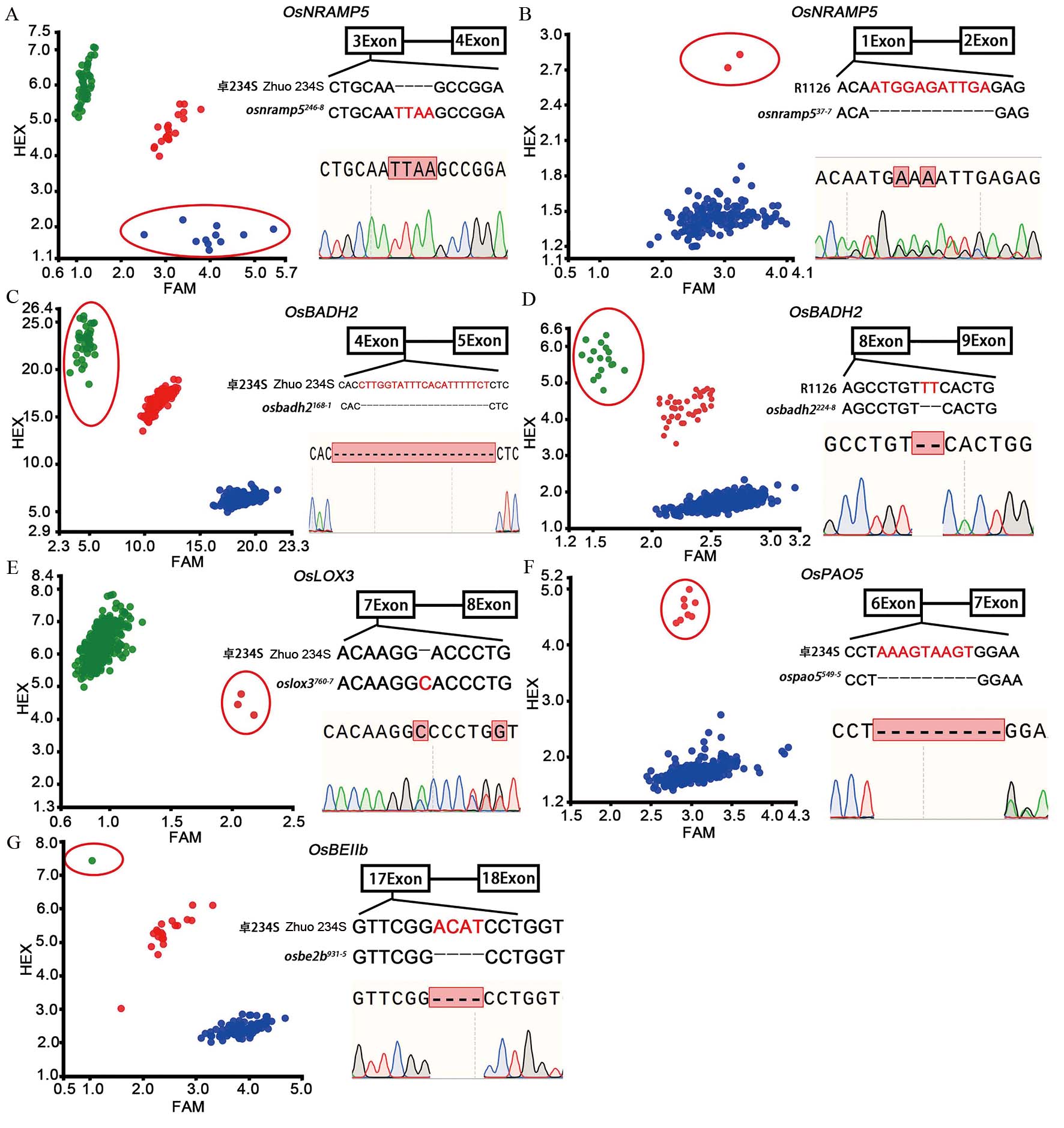
图2 M2群体的KASP鉴定及突变单株的Sanger测序验证 A, B:卓234S和湘农恢1126(R1126) M2群体OsNRAMP5基因KASP鉴定及突变单株Sanger测序验证,osnramp5246-8和osnramp537-7分别代表卓234S和湘农恢1126的OsNRAMP5基因突变单株; C, D:卓234S和湘农恢1126 M2群体OsBADH2基因KASP鉴定及Sanger测序验证,osbadh2168-1和 osbadh2224-8分别代表卓234S和湘农恢1126的OsBADH2基因突变单株;E~G:卓234S M2群体OsLOX3、OsPAO5和OsBEⅡb基因KASP鉴定及Sanger测序验证,oslox3760-7、ospao5549-5和osbe2b931-5分别代表卓234S的OsLOX3、OsPAO5和OsBEⅡb基因突变单株。图A中红点代表杂合基因型,蓝点代表纯合突变基因型,绿点代表野生基因型;图B~G中红点代表杂合基因型,绿点代表纯合突变基因型,蓝点代表野生基因型;红色椭圆形框内圆点代表目标基因纯合/杂合突变基因型。
Fig. 2. KASP identification of M2 generation and Sanger sequencing of mutant plants A and B, KASP identification of OsNRAMP5 gene in Zhuo 234S and Xiangnonghui 1126 M2 population and Sanger sequencing of mutants. osnramp5246-8 and osnramp537-7 represent mutant plant of OsNRAMP5 gene in Zhuo 234S and Xiangnonghui 1126, respectively; C and D, KASP identification of OsBADH2 gene in Zhuo 234S and Xiangnonghui 1126 M2 population and Sanger sequencing of mutants; osbadh2168-1and osbadh2224-8 represent mutant plant of OsBADH2 gene in Zhuo234S and Xiang Nonghui 1126, respectively. E-G, KASP identification of OsLOX3, OsPAO5 and OsBEⅡb gene in Zhuo 234S M2 population and Sanger sequencing of mutants. oslox3760-7, ospao5549-5 and osbe2b931-5 represented mutant plant of OsLOX3, OsPAO5 and OsBEⅡb gene of Zhuo 234S, respectively. In panel A, red dots represent heterozygous genotype, blue dots represent homozygous mutant genotype, green dots represent wild genotype. In panels B-G, red dots represent heterozygous genotype, green dots represent homozygous mutant genotype, blue dots represent wild genotype. The dots in the red oval box represent the homozygous/heterozygous genotype of target genes.
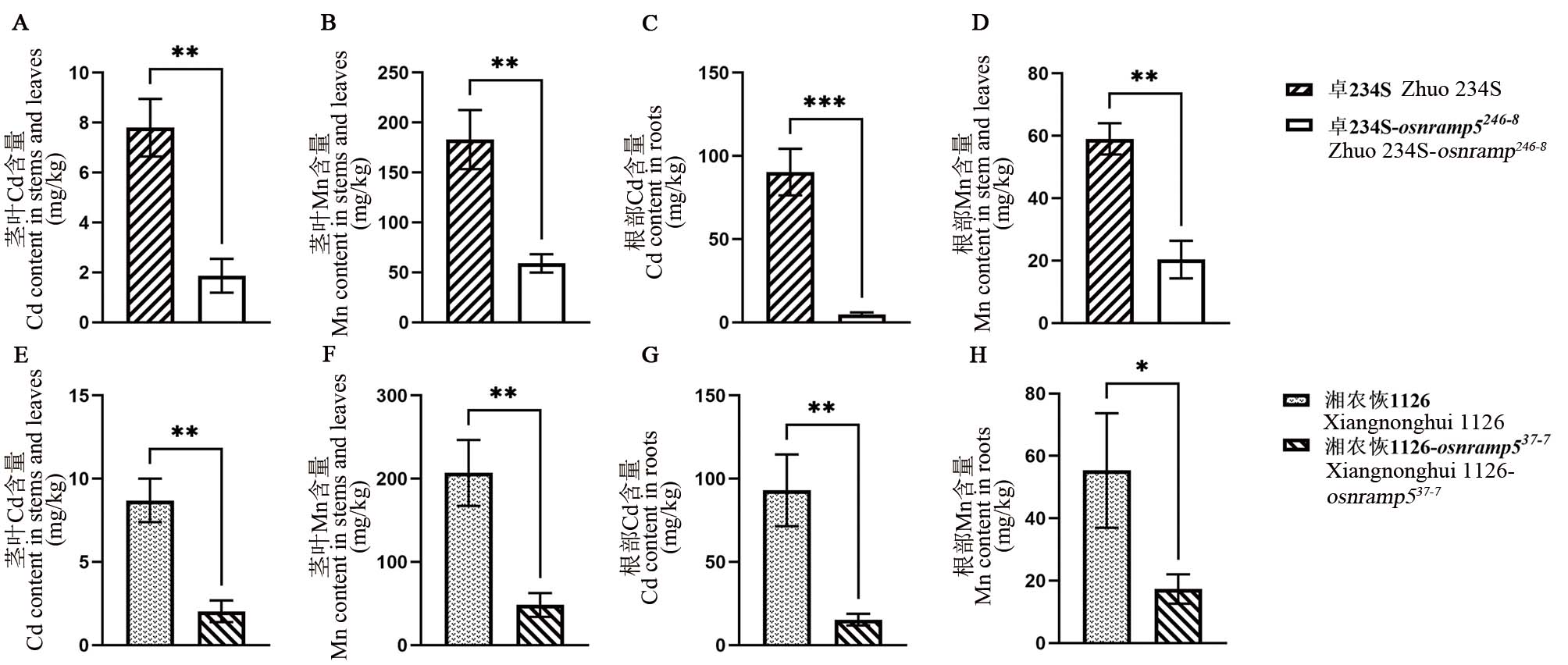
图3 OsNRAMP5纯合突变株及对照Cd、Mn含量 A-D: 纯合突变不育系卓234S-osnramp5246-8及原始对照材料Cd、Mn含量;E-H:纯合突变恢复系湘农恢1126-osnramp537-7及原始对照材料Cd、Mn含量;*、**和***分别表示在0.05、0.01和0.001水平上的差异显著(t检验)。
Fig. 3. Contents of Cd and Mn in homozygous mutant of OsNRAMP5 and control A-D, Contents of Cd and Mn in homozygous mutant Zhuo 234S-osnramp5246-8 and its original control material; E-H, Contents of Cd and Mn in the homozygous mutant Xiangnonghui 1126-osnramp537-7and its original control material; *, **, and *** represent difference was significant at 0.05, 0.01, and 0.001 levels, respectively (t-test).
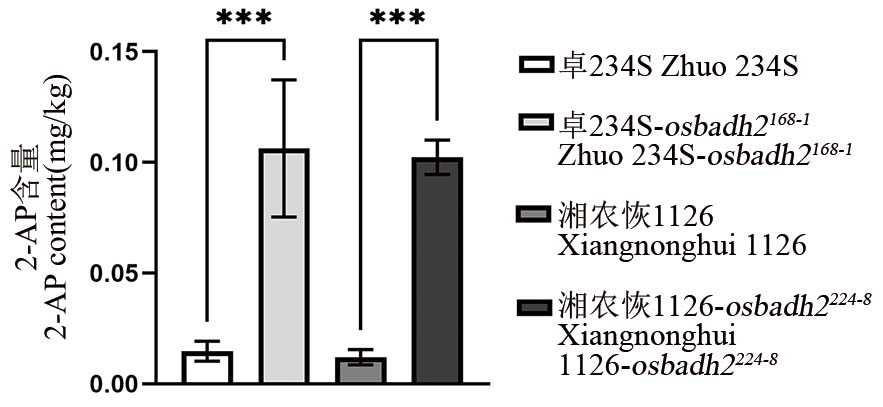
图4 OsBADH2纯合突变株及对照香味物质2-AP含量 ***表示在0.001水平上的差异(t检验)。
Fig. 4. 2-AP content in M2 homozygous mutant of OsBADH2 and control *** represent difference at the level of 0.001 (t-test).
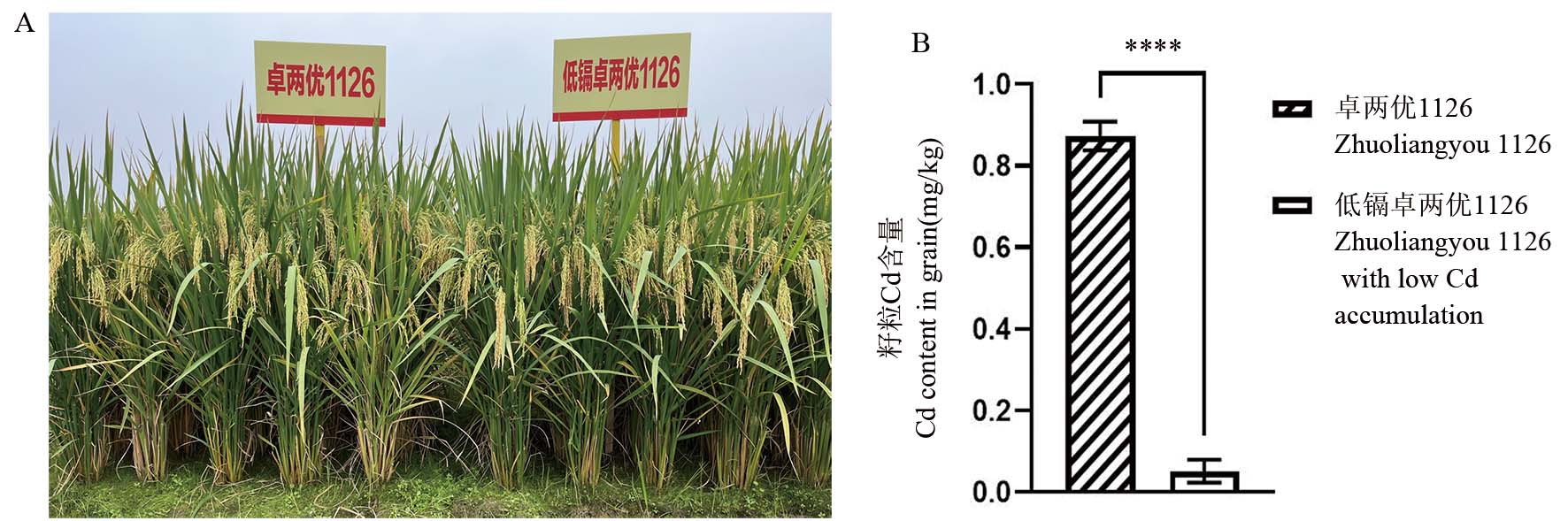
图5 镉低积累卓两优1126与原始对照品种田间表型及盆栽籽粒镉含量
Fig. 5. Field phenotype and cadmium content in grain of Zhuoliangyou 1126 with low cadmium accumulation compared with its original control
| 材料 Material | 株高 Plant height (cm) | 有效分蘖数 No. of effective tillers | 主穗长 Panicle length(cm) | 一次枝梗数 No. of primary branches | 二次枝梗数 No. of secondary branches |
|---|---|---|---|---|---|
| 卓两优1126(对照) Zhuoliangyou 1126(CK) | 124.6±3.3 a | 13.2±1.3 a | 28.9±1.4 a | 18.2±1.3 a | 99.4±6.4 a |
| 镉低积累卓两优1126 Zhuoliangyou 1126 with low cadmium accumulation | 124.8±2.7 a | 13.6±1.1 a | 27.9±0.9 a | 19.0±0.7 a | 104.2±5.7 a |
| 材料 Material | 总粒数 Spikelets per panicle | 实粒数 Grains per panicle | 结实率 Seed setting rate(%) | 千粒重 1000-grain weight(g) | |
| 卓两优1126(对照) Zhuoliangyou 1126(CK) | 347.4±39.1 a | 301.6±28.9 a | 87.0±2.0 a | 22.9±0.4 a | |
| 镉低积累卓两优1126 Zhuoliangyou 1126 with low cadmium accumulation | 360.8±24.1 a | 316.8±19.6 a | 88.0±2.0 a | 22.9±0.5 a |
表6 镉低积累卓两优1126及其原始对照品种农艺性状比较
Table 6. Comparison of agronomic traits of Zhuoliangyou 1126 with low cadmium accumulation and its original control varieties
| 材料 Material | 株高 Plant height (cm) | 有效分蘖数 No. of effective tillers | 主穗长 Panicle length(cm) | 一次枝梗数 No. of primary branches | 二次枝梗数 No. of secondary branches |
|---|---|---|---|---|---|
| 卓两优1126(对照) Zhuoliangyou 1126(CK) | 124.6±3.3 a | 13.2±1.3 a | 28.9±1.4 a | 18.2±1.3 a | 99.4±6.4 a |
| 镉低积累卓两优1126 Zhuoliangyou 1126 with low cadmium accumulation | 124.8±2.7 a | 13.6±1.1 a | 27.9±0.9 a | 19.0±0.7 a | 104.2±5.7 a |
| 材料 Material | 总粒数 Spikelets per panicle | 实粒数 Grains per panicle | 结实率 Seed setting rate(%) | 千粒重 1000-grain weight(g) | |
| 卓两优1126(对照) Zhuoliangyou 1126(CK) | 347.4±39.1 a | 301.6±28.9 a | 87.0±2.0 a | 22.9±0.4 a | |
| 镉低积累卓两优1126 Zhuoliangyou 1126 with low cadmium accumulation | 360.8±24.1 a | 316.8±19.6 a | 88.0±2.0 a | 22.9±0.5 a |
| [1] | Oladosu Y, Rafii M, Abdullah N, Hussin G, Ramli A, Rahim H, Miah G, Usman M. Principle and application of plant mutagenesis in crop improvement: A review[J]. Biotechnology & Biotechnological Equipment, 2016, 30(1): 1-16. |
| [2] | Sahu P, Sao R, Mondal S, Vishwakarma G, Gupta S, Kumar V, Singh S, Sharma D, Das B. Next generation sequencing based forward genetic approaches for identification and mapping of causal mutations in crop plants: A comprehensive review[J]. Plants (Basel), 2020, 9(10): 1355. |
| [3] | Bagher A, Nahid A, Mohsen M, Vahid M. Nuclear techniques in agriculture and genetics[J]. American Journal of Bioscience, 2014, 2(3): 102-105. |
| [4] | Jankowicz-Cieslak J, Tai T, Kumlehn J, Till B. Biotechnologies for plant mutation breeding: Protocols[M]. Springer Nature, 2017. |
| [5] | Spencer-Lopes M, Forster B, Jankuloski L. Manual on mutation breeding[M]. Food and Agriculture Organization of the United Nations (FAO), 2018. |
| [6] | Tokuyama Y, Furusawa Y, Ide H, Yasui A, Terato H. Role of isolated and clustered DNA damage and the post-irradiating repair process in the effects of heavy ion beam irradiation[J]. Journal of Radiation Research, 2015, 56(3): 446-455. |
| [7] | Hamada N, Imaoka T, Masunaga S, Ogata T, Okayasu R, Takahashi A, Kato T A, Kobayashi Y, Ohnishi T, Ono K, Shimada Y, Teshima T. Recent advances in the biology of heavy-ion cancer therapy[J]. Journal of Radiation Research, 2010, 51(4): 365-383. |
| [8] | Tanaka A, Shikazono N, Hase Y. Studies on biological effects of ion beams on lethality, molecular nature of mutation, mutation rate, and spectrum of mutation phenotype for mutation breeding in higher plants[J]. Journal of Radiation Research, 2010, 51(3): 223-233. |
| [9] | Hirano T, Kazama Y, Ishii K, Ohbu S, Shirakawa Y, Abe T. Comprehensive identification of mutations induced by heavy-ion beam irradiation in Arabidopsis thaliana[J]. Plant Journal, 2015, 82(1): 93-104. |
| [10] | Dong X, Yan X, Li W. Plant mutation breeding with heavy ion irradiation at IMP[J]. Journal of Agricultural Science, 2016, 8(5): 34-41. |
| [11] | Abe T, Kazama Y, Hirano T. Ion beam breeding and gene discovery for function analyses using mutants[J]. Nuclear Physics News, 2015, 25(4): 30-34. |
| [12] | 韶也, 彭彦, 毛毕刚, 余丽霞, 唐丽, 李曜魁, 胡远艺, 张丹, 袁智成, 罗武中, 彭选明, 李文建, 周利斌, 柏连阳, 赵炳然. M1TDS技术及镉低积累杂交水稻亲本创制与组合选育[J]. 杂交水稻, 2022, 37(1): 1-11. |
| Shao Y, Peng Y, Mao B G, Yu L X, Tang L, Li Y K, Hu Y Y, Zhang D, Yuan Z C, Luo W Z, Peng X M, Li W J, Zhou L B, Bai L Y, Zhao B R. M1TDS technology and breeding of Cd low-accumulating hybrid rice parents and combinations[J]. Hybrid Rice, 2022, 37(1): 1-11. (in Chinese with English abstract) | |
| [13] | 赵连芝, 王浩瀚, 王勇, 李雁民, 甄东升, 颉红梅. 重离子辐照选育春小麦新品种初探[J]. 西北农业学报, 2006(3): 17-19. |
| Zhao L Z, Wang H H, Wang Y, Li Y M, Zhen D S, Xie H M. Preliminary study on breeding new spring wheat varieties by heavy-ion irradiation[J]. Acta Agriculturae Boreali-occidentalis Sinica, 2006(3): 17-19. (in Chinese with English abstract) | |
| [14] | Melsen K, van de Wouw M, Contreras R. Mutation breeding in ornamentals[J]. HortScience, 2021, 56(10): 1154-1165. |
| [15] | Hu W, Li W, Chen J. Recent advances of microbial breeding via heavy-ion mutagenesis at IMP[J]. Letters in Applied Microbiology, 2017, 65(4): 274-280. |
| [16] | 陆栋, 王颖, 刘青芳, 吴鑫, 王菊芳, 马爽, 李文建. 12C离子束辐照对酵母发酵能力的影响[J]. 核技术, 2010, 33(5): 350-353. |
| Lu D, Wang Y, Liu Q F, Wu X, Wang J F, Ma S, Li W J. Effects of 12C ion beam irradiation on yeast fermentation capacity[J]. Nuclear Techniques, 2010, 33(5): 350-353. (in Chinese with English abstract) | |
| [17] | McCallum C M, Comai L, Greene E A, Henikoff S. Targeted screening for induced mutations[J]. Nature Biotechnology, 2000, 18(4): 455-7. |
| [18] | Colbert T, Till B, Tompa R, Reynolds S, Steine M, Yeung A, McCallum C, Comai L, Henikoff S. High-throughput screening for induced point mutations[J]. Plant Physiology, 2001, 126(2): 480-4. |
| [19] | Taheri S, Abdullah T, Jain S, Sahebi M, Azizi P. TILLING, high-resolution melting (HRM), and next-generation sequencing (NGS) techniques in plant mutation breeding[J]. Molecular Breeding, 2017, 37: 1-23. |
| [20] | Till B J, Datta S, Jankowicz-Cieslak J. TILLING: The next generation[J]. Advances in Biochemical Engineering-biotechnology, 2018, 164: 139-160. |
| [21] | Kumar A, McKeown P, Boualem A, Ryder P, Brychkova G, Bendahmane A, Sarkar A, Chatterjee M, Spillane C. TILLING by Sequencing (TbyS) for targeted genome mutagenesis in crops[J]. Molecular Breeding, 2017, 37: 1-12. |
| [22] | Kashtwari M, Wani A, Rather R. TILLING: An alternative path for crop improvement[J]. Journal of Crop Improvement, 2019, 33(1): 83-109. |
| [23] | Dai P, Wu L R, Chen S X, Wang M X, Cheng L Y, Zhang J X, Hao P, Yao W, Zarka J, Issa G C, Kwong L, Zhang D Y. Calibration-free NGS quantitation of mutations below 0.01% VAF[J]. Nature Communications, 2021, 12(1): 6123. |
| [24] | Song P, Chen S X, Yan Y H, Pinto A, Cheng L Y, Dai P, Patel A A, Zhang D Y. Selective multiplexed enrichment for the detection and quantitation of low-fraction DNA variants via low-depth sequencing[J]. Nature Biomedical Engineering, 2021, 5(7): 690-701. |
| [25] | Tang L, Mao B, Li Y, Lü Q, Zhang L, Chen C, He H, Wang W, Zeng X, Shao Y, Pan Y, Hu Y, Peng Y, Fu X, Li H, Xia S, Zhao B. Knockout of OsNramp5 using the CRISPR/Cas9 system produces low Cd-accumulating indica rice without compromising yield[J]. Scientific Reports, 2017, 7(1): 14438. |
| [26] | Hui S, Li H, Mawia AM, Zhou L, Cai J, Ahmad S, Lai C, Wang J, Jiao G, Xie L, Shao G, Sheng Z, Tang S, Wang J, Wei X, Hu S, Hu P. Production of aromatic three-line hybrid rice using novel alleles of BADH2[J]. Plant Biotechnology Journal, 2022, 20(1): 59-74. |
| [27] | Xu H, Wei Y, Zhu Y, Lian L, Xie H, Cai Q, Chen Q, Lin Z, Wang Z, Xie H, Zhang J. Antisense suppression of LOX3 gene expression in rice endosperm enhances seed longevity[J]. Plant Biotechnology Journal, 2015, 13(4): 526-39. |
| [28] | Lü Y, Shao G, Jiao G, Sheng Z, Xie L, Hu S, Tang S, Wei X, Hu P. Targeted mutagenesis of POLYAMINE OXIDASE 5 that negatively regulates mesocotyl elongation enables the generation of direct-seeding rice with improved grain yield[J]. Molecular Plant, 2021, 14(2): 344-351. |
| [29] | Sun Y, Jiao G, Liu Z, Zhang X, Li J, Guo X, Du W, Du J, Francis F, Zhao Y, Xia L. Generation of high-amylose rice through CRISPR/Cas9-mediated targeted mutagenesis of starch branching enzymes[J]. Frontiers in Plant Science, 2017, 8: 298. |
| [30] | Wang A, Jing Y, Cheng Q, Zhou H, Wang L, Gong W, Kou L, Liu G, Meng X, Chen M, Ma H, Shu X, Yu H, Wu D, Li J. Loss of function of SSIIIa and SSIIIb coordinately confers high RS content in cooked rice[J]. Proceedings of the National Academy of Sciences of the United States America, 2023, 120(19): e2220622120. |
| [31] | Kitamura S, Satoh K, Oono Y. Detection and characterization of genome-wide mutations in M1 vegetative cells of gamma-irradiated Arabidopsis[J]. PLoS Genetics, 2022, 18(1): e1009979. |
| [32] | Sasikala R, Kalaiyarasi R. Sensitivity of rice varieties to gamma irradiation[J]. Electronic Journal of Plant Breeding, 2010, 1(4): 845-889. |
| [33] | Gowthami R, Vanniarajan C, Souframanien J, Arumugam M. Comparison of radiosensitivity of two rice (Oryza sativa L.) varieties to gamma rays and electron beam in M1 generation[J]. Electronic Journal of Plant Breeding, 2017, 8(3): 732-741. |
| [34] | Yamaguchi H. Characteristics of ion beams as mutagens for mutation breeding in rice and chrysanthemums[J]. Japan Agricultural Research Quarterly, 2013, 47(4): 339-346. |
| [1] | 杨大兵, 杜雪树, 李进波, 夏明元, 胡亮, 石桓, 万丙良. 水稻抽穗期调控的分子机理及育种应用进展[J]. 中国水稻科学, 2026, 40(2): 145-154. |
| [2] | 倪晨, 张家豪, 朱昌进, 徐继伟, 胡秋倩, 霍中洋, 戴其根, 许轲, 李国辉. 水稻源流库形成与调控及其影响因素研究进展[J]. 中国水稻科学, 2026, 40(2): 155-170. |
| [3] | 王梦宁, 谢可冉, 高逖, 王真梅, 熊栋梁, 崔克辉. 高温对水稻粒重形成的影响及其栽培调控研究进展[J]. 中国水稻科学, 2026, 40(2): 171-180. |
| [4] | 罗肖郧, 郑兴飞, 彭宣国, 余启芝, 董华林, 殷得所, 王红波, 胡建林, 薛莲, 胡鹏, 徐得泽. 水稻抗倒伏研究:现状、挑战与未来方向[J]. 中国水稻科学, 2026, 40(2): 181-195. |
| [5] | 薛炮, 王友霜, 何弯弯, 黄晨博, 张涵, 丁震乾, 陈秋丽, 范运新, 丁成伟, 孙廉平, 胡婷婷. 水稻颖壳不闭合基因SG5的鉴定与克隆[J]. 中国水稻科学, 2026, 40(2): 210-222. |
| [6] | 段敏, 谢留杰, 岳雅妮, 黄善军. 基于CRISPR/Cas9技术创制优质香味粳稻品系[J]. 中国水稻科学, 2026, 40(2): 235-243. |
| [7] | 张梦柯, 陆佳雨, 何金, 许学, 吴爽, 王沛然, 陈若凡, 金青, 汪秀峰. 水稻野败型三系杂交种纯度检测功能标记的开发与应用[J]. 中国水稻科学, 2026, 40(2): 244-252. |
| [8] | 李兴沂, 陈玲, 邵建韬, 肖素勤, 李金璐, 付惠仙, 殷富有, 张建红, 程在全, 刘丽. 水稻产量与淀粉品质协同调控的分子遗传研究进展[J]. 中国水稻科学, 2026, 40(1): 1-17. |
| [9] | 岳轩宇, 谢文亚, 冯志明, 陈宗祥, 胡珂鸣, 左示敏. OsERF93参与调控水稻纹枯病抗性的研究[J]. 中国水稻科学, 2026, 40(1): 37-50. |
| [10] | 王轶欣, 林参, 马刘洋, 陈龙, 奉保华, 倪深, 魏祥进, 贺记外, 陈天晓. 谷丙转氨酶基因OsAlaAT4调控水稻氮素吸收和产量[J]. 中国水稻科学, 2026, 40(1): 51-60. |
| [11] | 黄奇娜, 姜鸿瑞, 杨婕, 于坤宇, 杨长登, 梁燕. 种子休眠基因Sdr4的生物信息学分析与分子标记开发和应用[J]. 中国水稻科学, 2026, 40(1): 61-71. |
| [12] | 程朝平, 何旎清, 白康呈, 林少俊, 黄凤凰, 刘军化, 程祖锌, 黄成志, 杨德卫. 聚合稻瘟病抗性基因Pigm-1和Pid2的水稻三系不育系福梦A的选育与利用[J]. 中国水稻科学, 2026, 40(1): 72-84. |
| [13] | 刘亚萍, 董译词, 郑君妍, 邱绚, 刘鹏程, 叶亚峰, 刘斌美, 陈析丰, 马伯军. 水稻类病变早衰突变体lmes7的鉴定与基因精细定位[J]. 中国水稻科学, 2026, 40(1): 85-94. |
| [14] | 谢世民, 周誉株, 薛晓迪, 朱广飞, 孙良, 陈建能. 水稻钵苗取栽协同作业式移栽机构设计与试验[J]. 中国水稻科学, 2026, 40(1): 131-144. |
| [15] | 王娟, 吴丽娟, 洪海波, 姚志文, 王磊, 鄂志国. 水稻泛素结合酶E2的生物学功能研究进展[J]. 中国水稻科学, 2025, 39(6): 744-750. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||