
中国水稻科学 ›› 2026, Vol. 40 ›› Issue (2): 196-209.DOI: 10.16819/j.1001-7216.2026.250411
杜志敏1,2,#, 贾一楠1,#, 段影青2, 曹妮2, 马刘洋2, 董欣丽2, 徐海1, 焦桂爱2, 唐绍清2,*( ), 胡培松2,*(
), 胡培松2,*( )
)
收稿日期:2025-04-24
修回日期:2025-06-03
出版日期:2026-03-10
发布日期:2026-03-16
通讯作者:
* email: tangshaoqing@caas.cn; peisonghu@126.com作者简介:#共同第一作者
基金资助:
DU Zhimin1,2,#, JIA Yinan1,#, DUAN Yingqing2, CAO Ni2, MA Liuyang2, DONG Xinli2, XU Hai1, JIAO Guiai2, TANG Shaoqing2,*( ), HU Peisong2,*(
), HU Peisong2,*( )
)
Received:2025-04-24
Revised:2025-06-03
Online:2026-03-10
Published:2026-03-16
Contact:
* email: tangshaoqing@caas.cn; peisonghu@126.comAbout author:#These authors contributed equally to the work;
摘要:
【目的】水稻种子休眠性变弱会导致收获前穗发芽(PHS),严重影响水稻的产量与品质,因而研究水稻种子休眠性的遗传调控机制至关重要。【方法】以中花11为背景通过CRISPR/Cas9技术创制水稻AP2/ERF转录因子OsERF34功能缺失突变体,结合转录组、激素代谢组及生理生化分析,解析其调控种子休眠的分子途径。【结果】发现oserf34突变体种子穗发芽严重,休眠性极显著减弱。转录组分析显示,oserf34与野生型之间差异表达基因(DEGs)显著富集于植物激素信号转导、ROS代谢及糖酵解通路,表明OsERF34可能通过调控激素合成与代谢、氧化还原平衡及糖代谢,维持种子休眠状态。进一步研究发现,oserf34突变体种子中生长素(IAA)、赤霉素(GA19/GA20)及水杨酸(SA)含量显著升高,而ROS清除剂脯氨酸含量降低,过氧化氢(H2O2)和超氧阴离子积累加剧,同时抗氧化酶(SOD、POD、CAT)活性上调。此外,突变体胚乳中可溶性糖含量和α-淀粉酶活性显著升高,为打破休眠提供能量基础。【结论】OsERF34通过参与激素合成与代谢、氧化还原平衡及糖代谢正向调控水稻种子休眠,为解析水稻种子休眠与萌发分子机制及创制抗穗发芽水稻新品种提供了新的理论依据与遗传资源。
杜志敏, 贾一楠, 段影青, 曹妮, 马刘洋, 董欣丽, 徐海, 焦桂爱, 唐绍清, 胡培松. AP2转录因子OsERF34正向调控水稻种子休眠[J]. 中国水稻科学, 2026, 40(2): 196-209.
DU Zhimin, JIA Yinan, DUAN Yingqing, CAO Ni, MA Liuyang, DONG Xinli, XU Hai, JIAO Guiai, TANG Shaoqing, HU Peisong. AP2 Transcription Factor OsERF34 Positively Regulates Seed Dormancy in Rice[J]. Chinese Journal OF Rice Science, 2026, 40(2): 196-209.
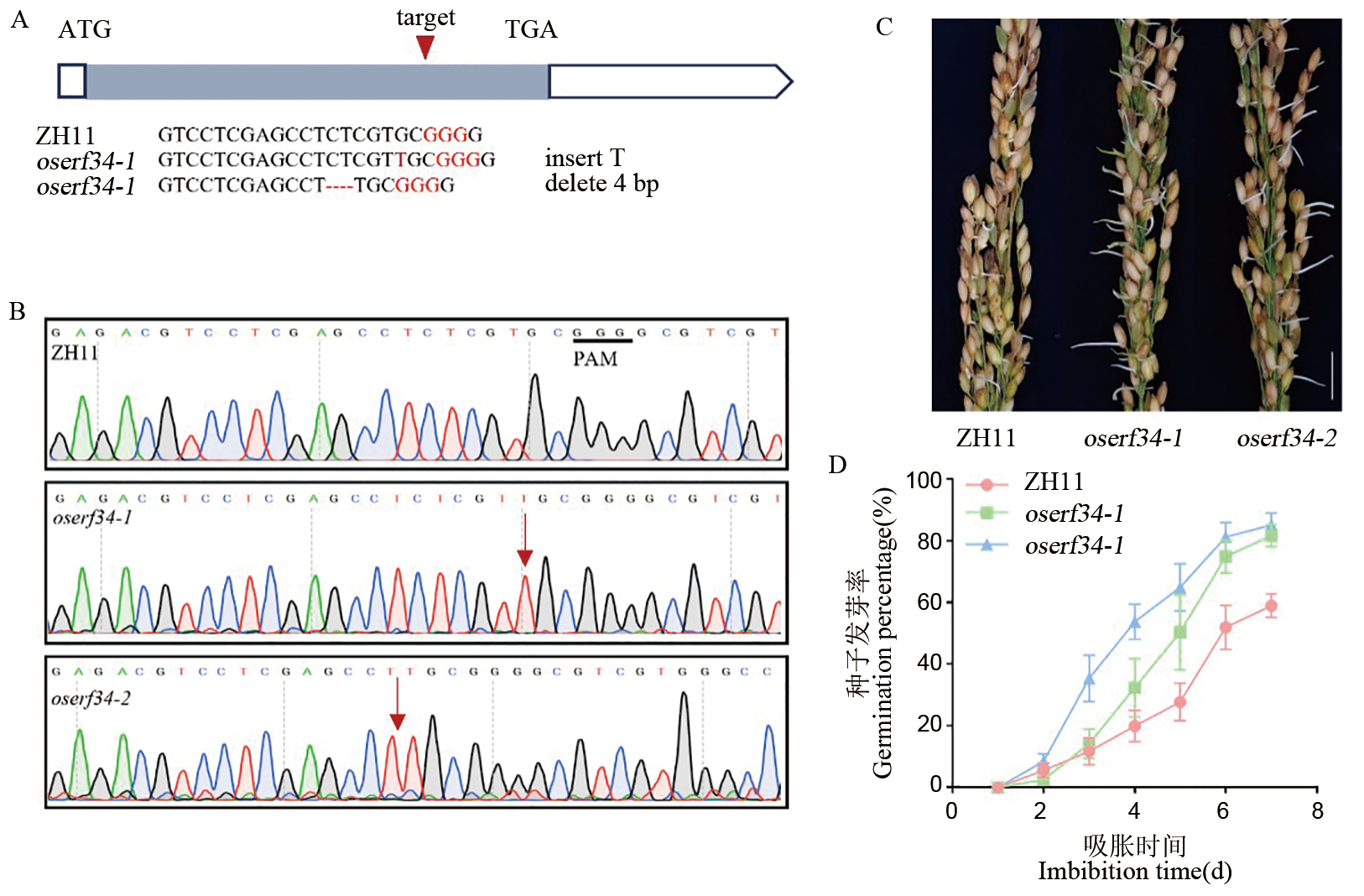
图1 oserf34突变体种子休眠性减弱 A:表示OsERF34 的基因结构、突变位点及突变方式,箭头表示CRISPR/Cas 9 编辑的靶位点,灰色矩形表示外显子,白色部分表示内含子。B:中花11靶位点序列和2种突变类型的突变信息及测序峰图。红色矩形位置是碱基插入和缺失的位置。C:收获授粉后30 d(DAP)的中花11和oserf34穗子,立即泡在ddH2O中,每24 h换一次水,C图为第4天的萌发表型。比例尺:2 cm。D:新鲜种子的萌发百分比随时间的变化。误差条表示标准差(n=3)。
Fig. 1. The oserf34 mutant exhibits a weaker dormancy phenotype A, Schematic diagram illustrating the gene structure of OsERF34, mutation sites, and mutation types. Arrows indicate CRISPR/Cas9 editing target sites. Gray rectangles represent exons, and white regions denote introns. B, Target site sequence in ZH11, mutation details of two mutation types, and sequencing chromatograms. Red rectangles highlight positions of nucleotide insertions and deletions (InDels). C, Panicles of ZH11 and oserf34 harvested at 30 days after pollination (DAP) were immediately soaked in ddH2O, with the water changed every 24 hours. The image shows the sprouting phenotype on the fourth day. Scale bar: 2 cm. D, Germination percentage of fresh seeds over time. Error bars represent standard deviation (n=3).
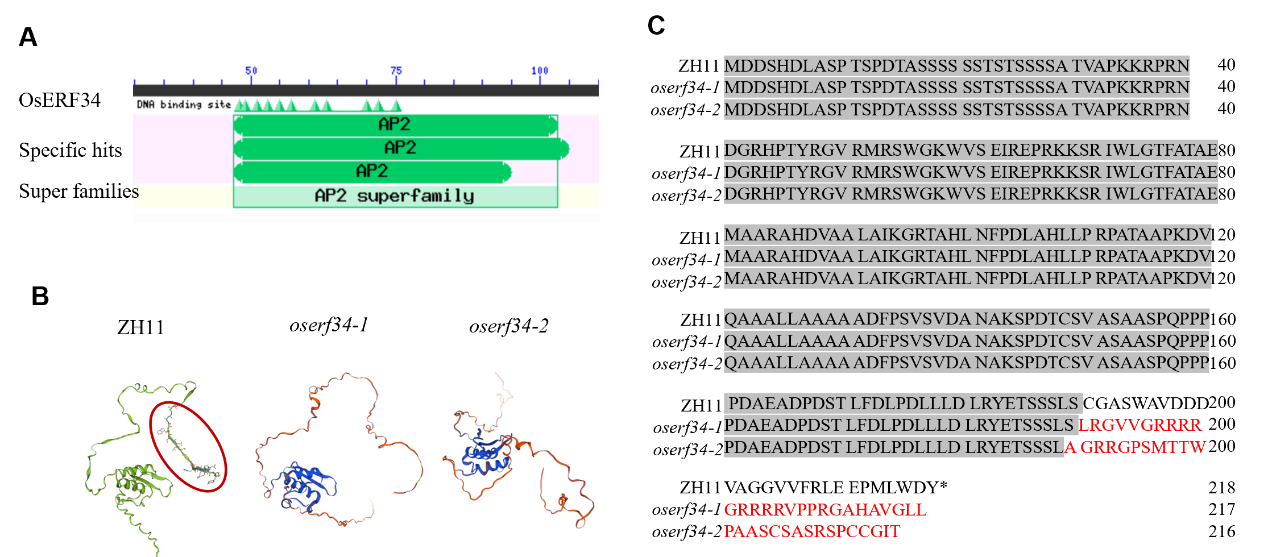
图2 OsERF34 的蛋白保守结构域及三维结构 A:NCBI网站分析OsERF34的蛋白保守结构域。B:SWISS-MODEL分析OsERF34突变前后的蛋白三级结构。红色圆圈表示变化的氨基酸位置。C:OsERF34突变后氨基酸序列的改变,红色字体表示变化的氨基酸。
Fig. 2. Analysis of conserved protein domains and tertiary structure in OsERF34 A, Conserved protein domains of OsERF34 identified using the NCBI database. B, Tertiary protein structures of OsERF34 before and after mutation analyzed using SWISS-MODEL. Red circles indicate positions of altered amino acids. C, Changes in the amino acid sequence of OsERF34 post-mutation. Amino acids modified by the mutation are highlighted in red.
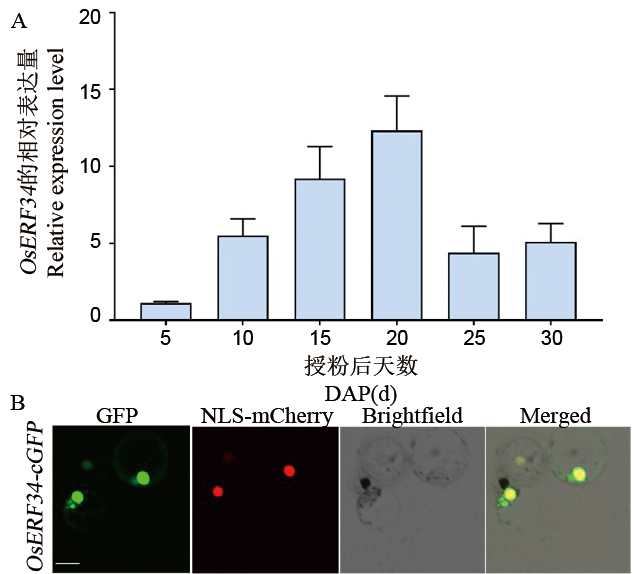
图3 OsERF34在不同发育时期种子中的表达模式及亚细胞定位 A:使用qRT-PCR 检测OsERF34在中花11不同授粉天数种子中的相对表达量表达量,以Ubi基因的表达量为内参进行标准化,将DAP5的表达量设为1,数值代表平均值±标准差,生物学重复3次。B:OsERF34-GFP 在水稻原生质体中的亚细胞定位。将OsERF34-GFP 和细胞核定位的Nuclear localization signal (NLS)-mCherry 共转进水稻原生质体。GFP,绿色荧光蛋白,标尺=10 μm。
Fig. 3. Expression patterns in seeds at different developmental stages of OsERF34 and its subcellular localization A, Relative expression levels of OsERF34 in Zhonghua11 seeds at different days after pollination (DAP) analyzed by qRT-PCR. Expression levels were normalized to the Ubi gene as an internal control, with the DAP5 value set to 1. Data represent mean ± standard deviation (n = 3). B, Subcellular localization of OsERF34-GFP in rice protoplasts. OsERF34-GFP and nuclear-localized NLS-mCherry were co-transfected into rice protoplasts. GFP, Green fluorescent protein. Scale bar: 10 μm.
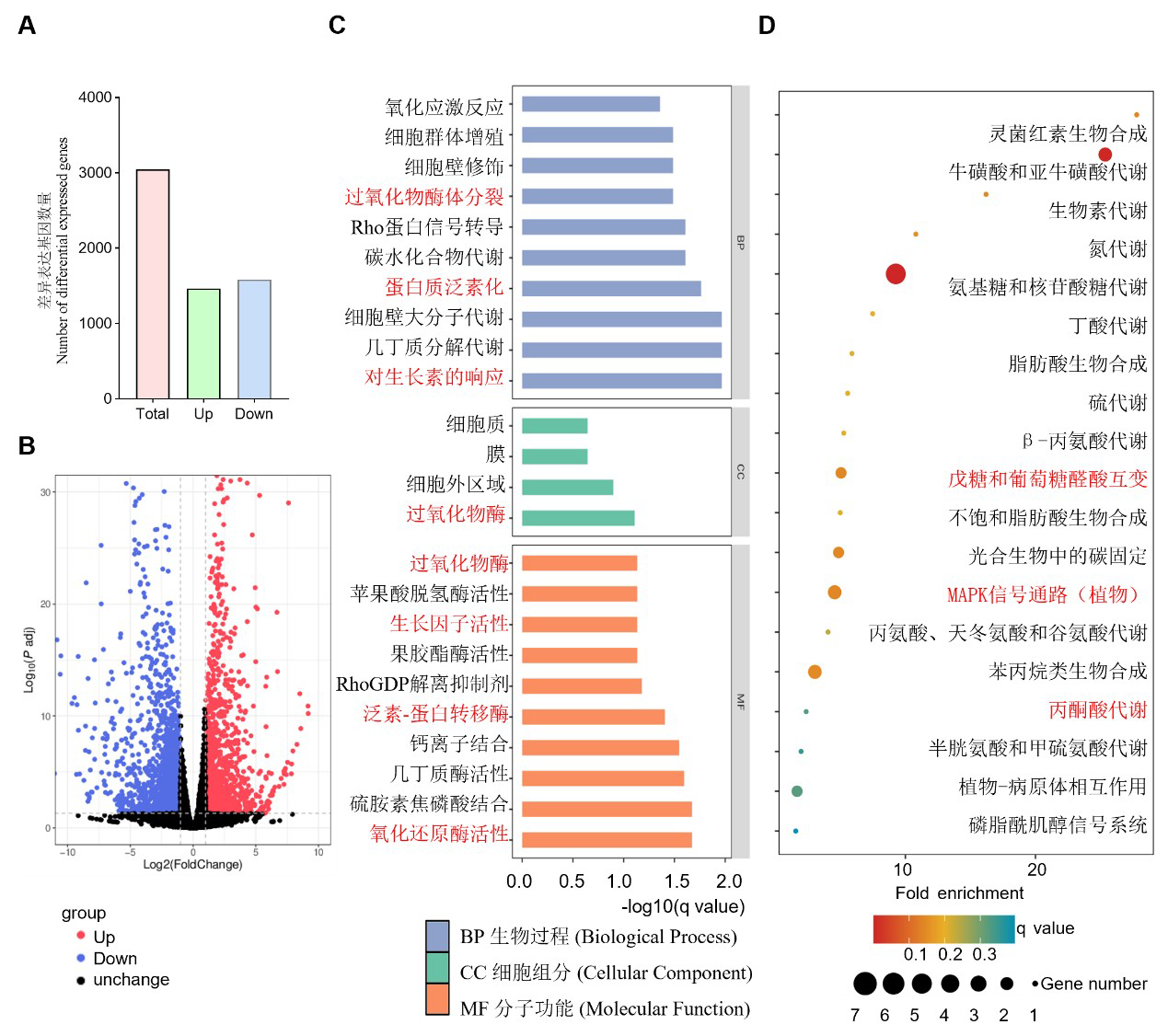
图4 Oserf34 和 中花11 的转录组测序分析 A: Oserf34与中花11差异表达基因的数目。B: oserf34与中花11授粉后20 d种子的总基因表达谱火山图。C: DEGs的GO富集中显著性排名前20的功能分析(Padj<0.05)。D: DEGs的KEGG富集中显著性排名前20的通路分析。
Fig. 4. Transcriptome sequencing analysis of oserf34 and Zhonghua11 A, Number of differentially expressed genes (DEGs) between oserf34 and Zhonghua11. B, Volcano plot of global gene expression profiles in seeds of oserf34 and Zhonghua11 at 20 days after pollination (DAP). C, Gene Ontology (GO) enrichment analysis of DEGs, showing the top 20 significantly enriched functional terms(Padj<0.05). D, KEGG pathway enrichment analysis of DEGs, displaying the top 20 significantly enriched pathways.
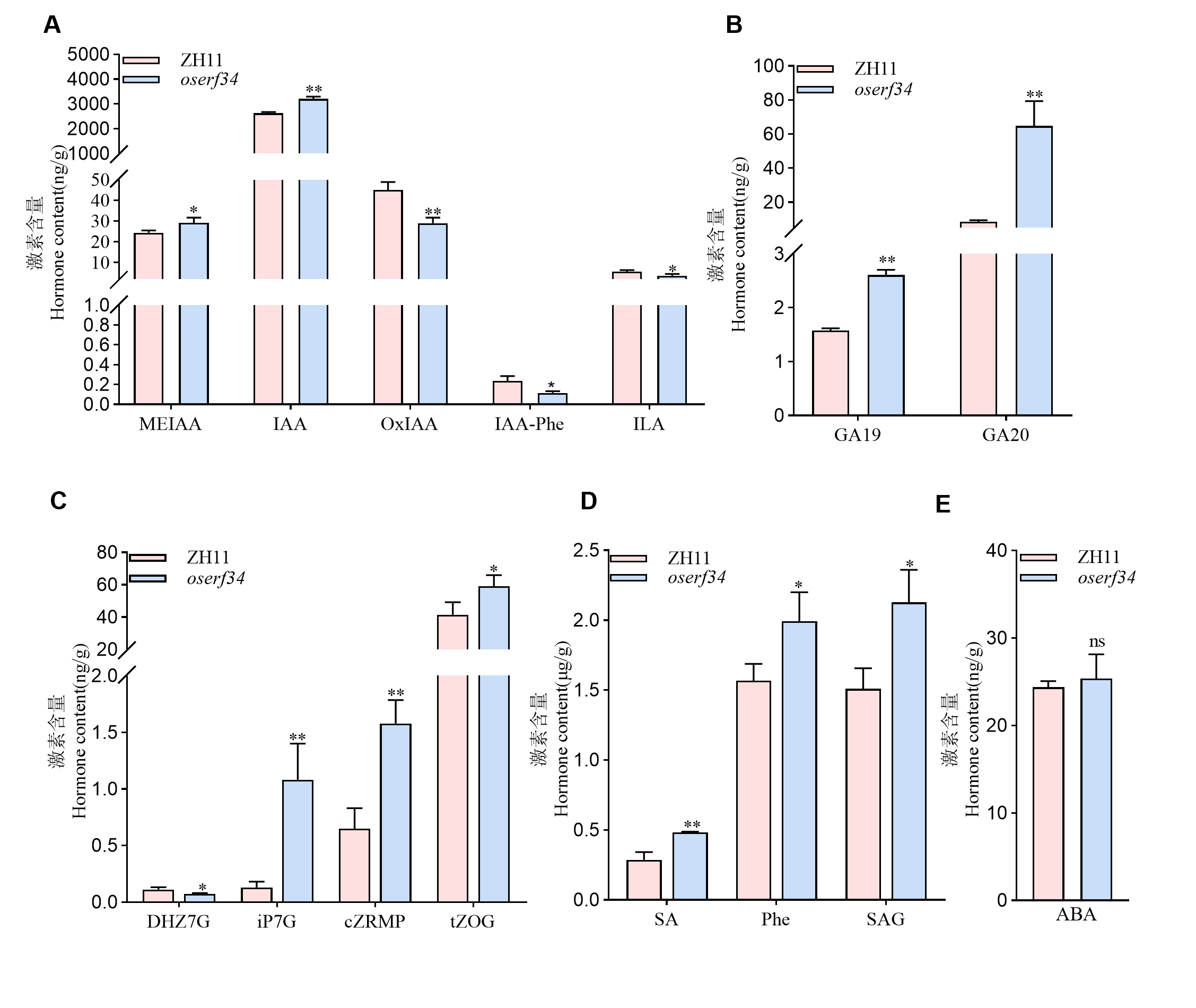
图5 中花11和oserf34激素含量的分析 A:授粉后25 d 中花11和oserf34中生长素类激素的含量。MEIAA:吲哚-3-乙酸甲酯;IAA:生长素;OxIAA:氧化吲哚乙酸;IAA-Phe:N-(3-吲哚乙酰基)-L-苯丙氨酸;ILA:吲哚-3-乳酸。B,授粉后25 d 中花11和oserf34赤霉素类激素的含量。GA19:赤霉素19;GA20:赤霉素20。C,授粉后25 d 中花11和oserf34细胞分裂素类激素的含量。DHZ7G:双氢玉米素-7-糖苷;iP7G:异戊烯腺嘌呤-7-葡糖苷;cZRMP:顺式玉米素-糖苷磷酸盐;tZOG:反式-玉米素-9-Β-葡萄糖苷。D:授粉后25 d 中花11和oserf34水杨酸类激素的含量。SA:水杨酸;Phe:L-苯丙氨酸;SAG:水杨酸-2-O-β-葡萄糖苷。数值代表平均值±标准差(n=3)。E:授粉后25 d 中花11和oserf34中脱落酸(ABA)含量。野生型中花11和突变体oserf34之间进行差异比较分析,**代表在P <0.01水平上存在显著差异, *代表在P <0.05水平上存在显著差异(学生氏t检验)。
Fig. 5. Analysis of hormone contents between Zhonghua11 and oserf34 A, Auxin content in Zhonghua11 and oserf34 at 25 days after pollination. MEIAA, Methyl indole-3-acetate; IAA, Indole-3-acetic acid; OxIAA, 2-oxindole-3-acetic acid; IAA-Phe, N-(3-Indolylacetyl)-L-phenylalanine; ILA, Indole-3-lactic acid. B, Gibberellin content in Zhonghua11 and oserf34 at 25 days after pollination. GA19, Gibberellin A19; GA20, Gibberellin A20. C, Cytokinin content in Zhonghua11 and oserf34 at 25 days after pollination. DHZ7G, Dihydrozeatin-7-glucoside; iP7G, N6-Isopentenyl-adenine-7-glucoside; cZRMP, cis-Zeatin riboside monophosphate; tZOG, trans-Zeatin-O-glucoside. D, Salicylic acid content in Zhonghua 11 and oserf34 at 25 days after pollination. SA, Salicylic acid; Phe, L-Phenylalanine; SAG, Salicylic acid 2-O-β-glucoside. E, ABA content in Zhonghua 11 and oserf34. Differential comparative analysis between Zhonghua 11 and oserf34, error bars represent SD (n = 3). **, P < 0.01; *, P < 0.05(Student’s t-test).
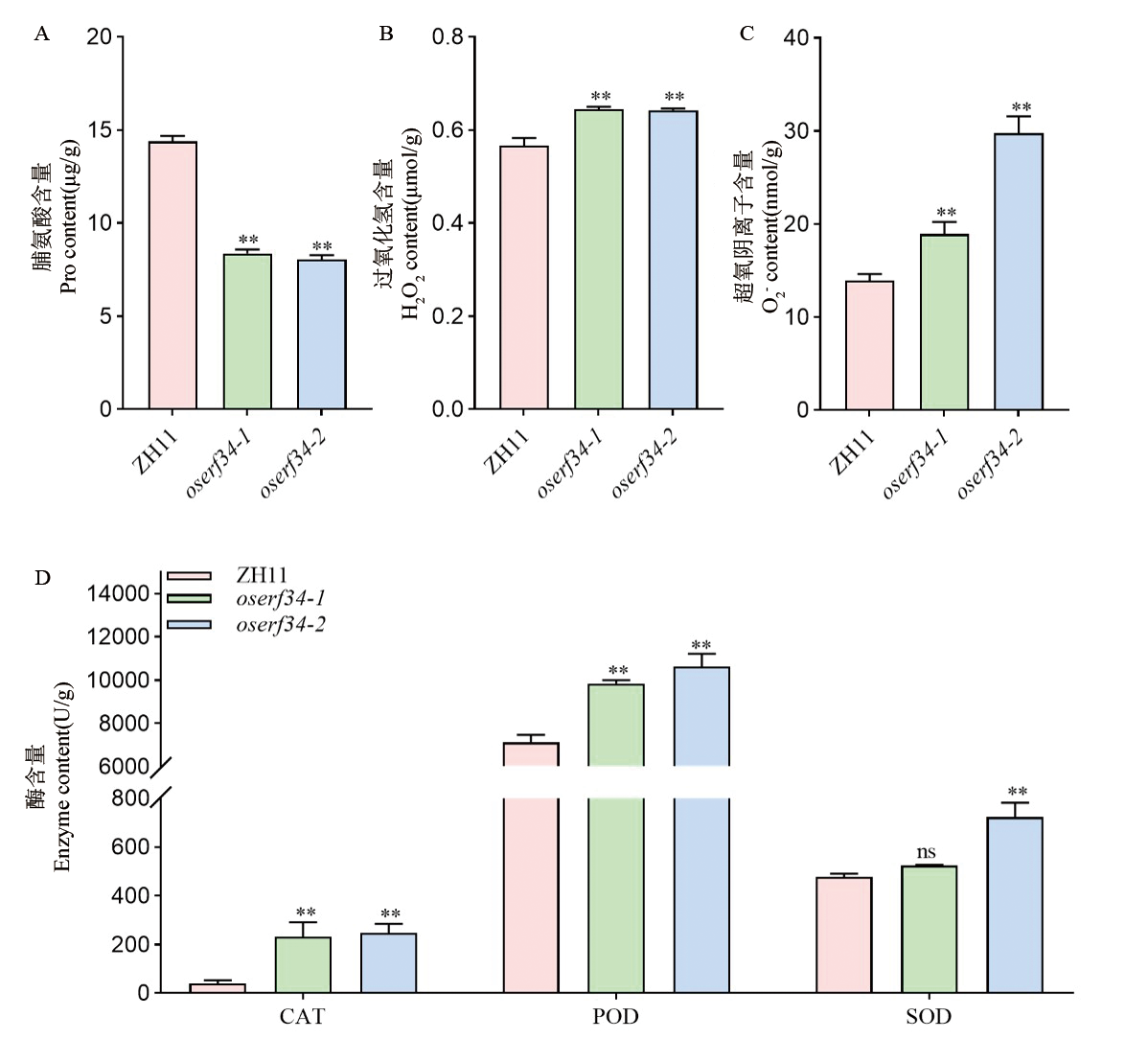
图6 ZH11和oserf34活性氧途径分析 A:ZH11和oserf34 授粉后20 d的种子中脯氨酸含量。B:ZH11和oserf34授粉后20 d的种子中过氧化氢含量。C:ZH11和oserf34授粉后20 d的种子中超氧阴离子含量。D:ZH11和oserf34授粉后20 d的种子中ROS清除酶含量。数值代表平均值±标准差(n=3)。野生型ZH11和突变体oserf34之间进行差异比较分析,**代表在P <0.01水平上存在显著差异, *代表在P <0.05水平上存在显著差异,t检验。
Fig. 6. Analysis of reactive oxygen species pathways between ZH11 and oserf34 A, Determination of proline content in seeds of ZH11 and oserf34 at 20 days after pollination (DAP). B, Determination of hydrogen peroxide (H2O2) content in seeds of ZH11 and oserf34 at 20 DAP. C, Determination of superoxide anion content in seeds of ZH11 and oserf34 at 20 DAP. D, Determination of ROS-scavenging enzyme activities in seeds of ZH11 and oserf34 at 20 DAP. Differential comparative analysis between wild-type ZH11 and mutant oserf34, error bars represent SD (n = 3). **, P < 0.01; *, P < 0.05, Student’s t-test.
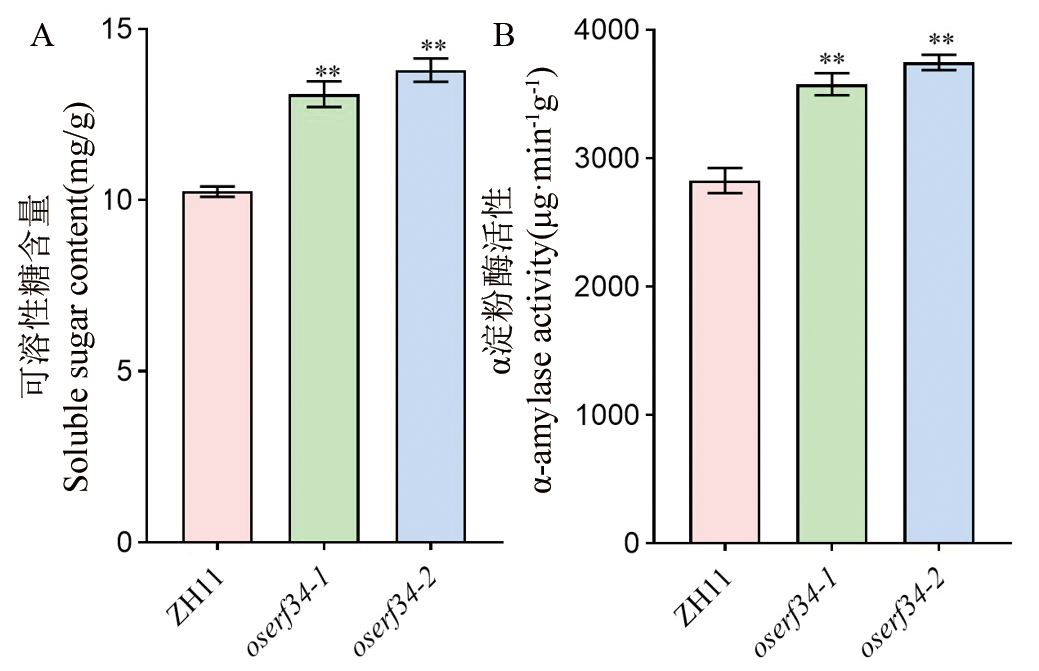
图7 ZH11和oserf34糖代谢相关指标分析 A:授粉后20 d的种子中可溶性糖含量的测定。B:授粉后20 d的种子中α-淀粉酶活性的测定。数值代表平均值±标准差(n=3)。**代表野生型ZH11和突变体oserf34在P <0.01水平上存在显著差异, *代表在P <0.05水平上存在显著差异(t检验)。
Fig. 7. Analysis of sugar metabolism-related parameters in ZH11 and oserf34 A, Measurement of soluble sugar content in seeds at 20 days after pollination (DAP). B, Measurement of α-amylase activity in seeds at 20 days after pollination (DAP). Differential comparative analysis between ZH11 and oserf34, error bars represent SD (n = 3). **, P < 0.01; *, P < 0.05 (Student’s t-test).
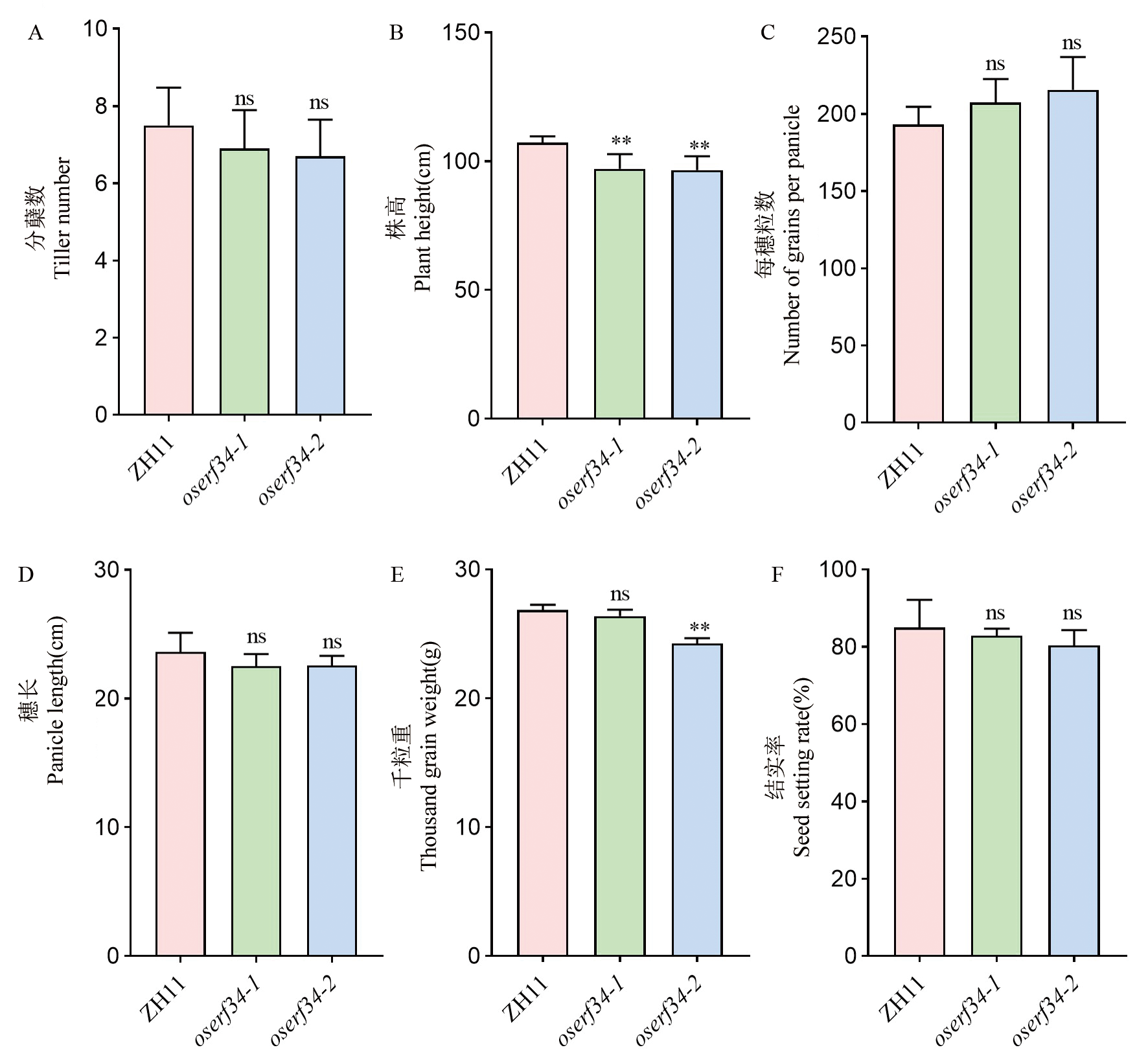
图8 田间农艺性状统计 A-D:ZH11和oserf34分蘖数(A),株高(B),每穗粒数(C)和穗长(D)。柱状图代表平均值±标准差(n=10)。E-F:千粒重(E)和结实率(F)。柱状图代表平均值±标准差(n=3)。**代表野生型ZH11和突变体oserf34在P<0.01水平上存在显著差异, ns代表无显著差异(t检验)。
Fig. 8. Statistical analysis of agronomic traits in field conditions A-D, Tiller number(A), plant height(B), grains per panicle(C), and panicle length(D) of ZH11 and oserf34. Bar graphs represent mean±SD(n=10). E-F, 1000-grain weight (E) and seed setting rate(F). Values are means±SD(n=3). ** and "ns" indicate extremely significant differences (Student’s t-test, P<0.01) and no significant difference between ZH11 and oserf34, respectively.
| [1] | Muthayya S, Sugimoto J D, Montgomery S, Maberly G F. An overview of global rice production, supply, trade, and consumption[J]. Annals of the New York Academy of Sciences, 2014, 1324: 7-14. |
| [2] | 储零逸. AP2/ERF转录因子OsERF44调控水稻穗发芽的效应及其机制研究[D]. 扬州: 扬州大学, 2021. |
| Chu L Y. Effect and mechanism of AP2/ERF transcription factor OsERF44 on panicle sprouting in rice[D]. Yangzhou: Yangzhou University, 2021. (in Chinese with English abstract) | |
| [3] | 左志梅, 赵凯, 朱海山. 外源ABA对不同品种番茄种子萌发的影响[J]. 长江蔬菜, 2014(18): 35-38. |
| Zuo Z M, Zhao K, Zhu H S. Effects of exogenous abscisic acid on germination of different tomato cultivar seeds[J]. Journal of Changjiang Vegetables, 2014(18): 35-38. (in Chinese with English abstract) | |
| [4] | Nonogaki H, Bassel G W, Bewley J D. Germination: Still a mystery[J]. Plant Science, 2010, 179(6): 574-581. |
| [5] | Bewley J D. Seed germination and dormancy[J]. The Plant Cell, 1997: 1055-1066. |
| [6] | Graeber K, Nakabayashi K, Miatton E, Leubner-Metzger G, Soppe W J J. Molecular mechanisms of seed dormancy[J]. Plant, Cell & Environment, 2012, 35(10): 1769-1786. |
| [7] | Nonogaki H. Seed dormancy and germination emerging mechanisms and new hypotheses[J]. Frontiers in Plant Science, 2014, 5: 233. |
| [8] | Finkelstein R, Reeves W, Ariizumi T, Steber C. Molecular aspects of seed dormancy[J]. Annual Review of Plant Biology, 2008, 59: 387-415. |
| [9] | Sutoh K, Washio K, Imai R, Wada M, Nakai T, Yamauchi D. An N-terminal region of a Myb-like protein is involved in its intracellular localization and activation of a gibberellin-inducible proteinase gene in germinated rice seeds[J]. Bioscience, Biotechnology, and Biochemistry, 2015, 79(5): 747-759. |
| [10] | Finch-Savage W E, Leubner-Metzger G. Seed dormancy and the control of germination[J]. New Phytologist, 2006, 171(3): 501-523. |
| [11] | Del Bel Z, Andrade A, Lindström L, Alvarez D, Vigliocco A, Alemano S. The role of the sunflower seed coat and endosperm in the control of seed dormancy and germination: Phytohormone profile and their interaction with seed tissues[J]. Plant Growth Regulation, 2024, 102(1): 51-64. |
| [12] | 陈兵先, 张琪, 戴彰言, 周旭, 刘军. 水杨酸引发提高低温下水稻种子萌发活力的生理与分子效应[J]. 中国农业科学, 2024, 57(7): 1220-1236. |
| Chen B X, Zhang Q, Dai Z Y, Zhou X, Liu J. Physiological and molecular effects of salicylic acid on rice seed germination at low temperature[J]. Scientia Agricultura Sinica, 2024, 57(7): 1220-1236. (in Chinese with English abstract) | |
| [13] | Nguyen T N, Tuan P A, Ayele B T. Jasmonate regulates seed dormancy in wheat via modulating the balance between gibberellin and abscisic acid[J]. Journal of Experimental Botany, 2022, 73(8): 2434-2453. |
| [14] | Shu K, Zhang H, Wang S, Chen M, Wu Y, Tang S, Liu C, Feng Y, Cao X, Xie Q. ABI4 regulates primary seed dormancy by regulating the biogenesis of abscisic acid and gibberellins in Arabidopsis[J]. PLoS Genetics, 2013, 9(6): e1003577. |
| [15] | Sun M, Shen Y, Chen Y, Wang Y, Cai X, Yang J, Jia B, Dong W, Chen X, Sun X. Osa-miR1320 targets the ERF transcription factor OsERF096 to regulate cold tolerance via JA-mediated signaling[J]. Plant Physiology, 2022, 189(4): 2500-2516. |
| [16] | To A, Valon C, Savino G, Guilleminot J, Devic M, Giraudat J, Parcy F. A network of local and redundant gene regulation governs Arabidopsis seed maturation[J]. The Plant Cell, 2006, 18(7): 1642-1651. |
| [17] | Xie Z, Zhang Z L, Hanzlik S, Cook E, Shen Q J. Salicylic acid inhibits gibberellin-induced alpha-amylase expression and seed germination via a pathway involving an abscisic-acid-inducible WRKY gene[J]. Plant Molecular Biology, 2007, 64(3): 293-303. |
| [18] | Anand A, Kumari A, Thakur M, Koul A. Hydrogen peroxide signaling integrates with phytohormones during the germination of magnetoprimed tomato seeds[J]. Scientific Reports, 2019, 9(1): 8814. |
| [19] | Guha T, Das H, Mukherjee A, Kundu R. Elucidating ROS signaling networks and physiological changes involved in nanoscale zero valent iron primed rice seed germination sensu stricto[J]. Free Radical Biology & Medicine, 2021, 171: 11-25. |
| [20] | Howard T P, Fahy B, Craggs A, Mumford R, Leigh F, Howell P, Greenland A, Smith A M. Barley mutants with low rates of endosperm starch synthesis have low grain dormancy and high susceptibility to preharvest sprouting[J]. The New Phytologist, 2012, 194(1): 158-167. |
| [21] | Li W Y, Chen B X, Chen Z J, Gao Y T, Chen Z, Liu J. Reactive oxygen species generated by NADPH oxidases promote radicle protrusion and root elongation during rice seed germination[J]. International Journal of Molecular Sciences, 2017, 18(1): 110. |
| [22] | 邓本良. 活性氧调控作物种子发芽机理的研究进展[J]. 安徽农业科学, 2020, 48(13): 21-22, 26. |
| Deng B L. Research advances of reactive oxygen species in regulating crop seed germination[J]. Journal of Anhui Agricultural Sciences, 2020, 48(13): 21-22, 26. (in Chinese with English abstract) | |
| [23] | Xiong M, Yu J, Wang J, Gao Q, Huang L, Chen C, Zhang C, Fan X, Zhao D, Liu Q Q, Li Q F. Brassinosteroids regulate rice seed germination through the BZR1-RAmy3D transcriptional module[J]. Plant Physiology, 2022, 189(1): 402-418. |
| [24] | Du L, Xu F, Fang J, Gao S, Tang J, Fang S, Wang H, Tong H, Zhang F, Chu J, Wang G, Chu C. Endosperm sugar accumulation caused by mutation of PHS8/ISA1 leads to pre-harvest sprouting in rice[J]. The Plant Journal, 2018, 95(3): 545-556. |
| [25] | Zhao J, Wang J, Liu J, Zhang P, Kudoyarova G, Liu C J, Zhang K. Spatially distributed cytokinins: Metabolism, signaling, and transport[J]. Plant Communications, 2024, 5(7): 100936. |
| [26] | Faix B, Radchuk V, Nerlich A, Hümmer C, Radchuk R, Neil Emery R J, Keller H, Götz K P, Weschke W, Geigenberger P, Weber H. Barley grains, deficient in cytosolic small subunit of ADP-glucose pyrophosphorylase, reveal coordinate adjustment of C: N metabolism mediated by an overlapping metabolic- hormonal control[J]. The Plant Journal, 2012, 69(6): 1077-1093. |
| [27] | Sugimoto K, Takeuchi Y, Ebana K, Miyao A, Hirochika H, Hara N, Ishiyama K, Kobayashi M, Ban Y, Hattori T, Yano M. Molecular cloning of Sdr4, a regulator involved in seed dormancy and domestication of rice[J]. Proceedings of the National Academy of Sciences of the United States of America, 2010, 107(13): 5792-5797. |
| [28] | Zhao B, Zhang H, Chen T, Ding L, Zhang L, Ding X, Zhang J, Qian Q, Xiang Y. Sdr4 dominates pre-harvest sprouting and facilitates adaptation to local climatic condition in Asian cultivated rice[J]. Journal of Integrative Plant Biology, 2022, 64(6): 1246-1263. |
| [29] | Song S, Wang G, Wu H, Fan X, Liang L, Zhao H, Li S, Hu Y, Liu H, Ayaad M, Xing Y. OsMFT2 is involved in the regulation of ABA signaling-mediated seed germination through interacting with OsbZIP23/66/72 in rice[J]. The Plant Journal, 2020, 103(2): 532-546. |
| [30] | Shen J, Zhang L, Wang H, Guo J, Li Y, Tan Y, Shu Q, Qian Q, Yu H, Chen Y, Song S. The phosphatidylethanolamine-binding proteins OsMFT1 and OsMFT2 regulate seed dormancy in rice[J]. The Plant Cell, 2024, 36(9): 3857-3874. |
| [31] | Xu F, Tang J, Wang S, Cheng X, Wang H, Ou S, Gao S, Li B, Qian Y, Gao C, Chu C. Antagonistic control of seed dormancy in rice by two bHLH transcription factors[J]. Nature Genetics, 2022, 54(12): 1972-1982. |
| [32] | Fu F F, Xue H W. Coexpression analysis identifies Rice Starch Regulator1, a rice AP2/EREBP family transcription factor, as a novel rice starch biosynthesis regulator[J]. Plant Physiology, 2010, 154(2): 927-938. |
| [33] | Gu C, Guo Z H, Hao P P, Wang G M, Jin Z M. Multiple regulatory roles of AP2/ERF transcription factor in angiosperm[J]. Botanical Studies, 2017, 58(1): 6. |
| [34] | Ma Z, Hu L, Jiang W. Understanding AP2/ERF transcription factor responses and tolerance to various abiotic stresses in plants: A comprehensive review[J]. International Journal of Molecular Sciences, 2024, 25(2): 893. |
| [35] | Ma Z, Wu T, Huang K, Jin Y M, Li Z, Chen M, Yun S, Zhang H, Yang X, Chen H, Bai H, Du L, Ju S, Guo L, Bian M, Hu L, Du X. A novel AP2/ERF transcription factor, OsRPH1, negatively regulates plant height in rice[J]. Frontiers in Plant Science, 2020, 11: 709. |
| [36] | Yaish M W, El-Kereamy A, Zhu T, Beatty P H, Good A G, Bi Y M, Rothstein S J. The APETALA-2-like transcription factor OsAP2-39 controls key interactions between abscisic acid and gibberellin in rice[J]. PLoS Genetics, 2010, 6(9): e1001098. |
| [37] | Qi W, Sun F, Wang Q, Chen M, Huang Y, Feng Y Q, Luo X, Yang J. Rice ethylene-response AP2/ERF factor OsEATB restricts internode elongation by down-regulating a gibberellin biosynthetic gene[J]. Plant Physiology, 2011, 157(1): 216-228. |
| [38] | Huang K, Lu F, Chen P, Jiao G, Lin H, Zhang J, Zhao S, Cao R, Shao G, Sheng Z, Hu S, Tang S, Hu P, Wei X. A large-scale gene regulatory network for rice endosperm starch biosynthesis and its application in genetic improvement of rice quality[J]. Plant Biotechnology Journal, 2025, 23(7): 2583-2594. |
| [39] | Huijser C, Kortstee A, Pego J, Weisbeek P, Wisman E, Smeekens S. The Arabidopsis sucrose uncoupled-6 gene is identical to ABSCISIC acid insensitive-4: Involvement of abscisic acid in sugar responses[J]. The Plant Journal, 2000, 23(5): 577-585. |
| [40] | Wang C, Wang H, Zhang J, Chen S. A seed-specific AP2-domain transcription factor from soybean plays a certain role in regulation of seed germination[J]. Science in China Series C: Life Sciences, 2008, 51(4): 336-345. |
| [41] | Arenas-Huertero F, Arroyo A, Zhou L, Sheen J, León P. Analysis of Arabidopsis glucose insensitive mutants, gin5 and gin6, reveals a central role of the plant hormone ABA in the regulation of plant vegetative development by sugar[J]. Genes & Development, 2000, 14(16): 2085-2096. |
| [42] | Shu K, Chen Q, Wu Y, Liu R, Zhang H, Wang P, Li Y, Wang S, Tang S, Liu C, Yang W, Cao X, Serino G, Xie Q. ABI4 mediates antagonistic effects of abscisic acid and gibberellins at transcript and protein levels[J]. The Plant Journal, 2016, 85(3): 348-361. |
| [43] | Tuan P A, Kumar R, Rehal P K, Toora P K, Ayele B T. Molecular mechanisms underlying abscisic acid/ gibberellin balance in the control of seed dormancy and germination in cereals[J]. Frontiers in Plant Science, 2018, 9: 668. |
| [44] | 刘建利. 7种激素对蒺藜状苜蓿种子发芽的影响[J]. 干旱地区农业研究, 2010, 28(2): 113-117. |
| Liu J L. Effects of 7 plant growth regulators on seed germination of Medicago truncatula[J]. Agricultural Research in the Arid Areas, 2010, 28(2): 113-117. (in Chinese with English abstract) | |
| [45] | Brady S M, Sarkar S F, Bonetta D, McCourt P. The abscisic acid insensitive 3 (ABI3) gene is modulated by farnesylation and is involved in auxin signaling and lateral root development in Arabidopsis[J]. The Plant Journal, 2003, 34(1): 67-75. |
| [46] | Liu P P, Montgomery T A, Fahlgren N, Kasschau K D, Nonogaki H, Carrington J C. Repression of AUXIN RESPONSE FACTOR10 by microRNA160 is critical for seed germination and post-germination stages[J]. The Plant Journal, 2007, 52(1): 133-146. |
| [47] | Ramaih S, Guedira M, Paulsen G M. Relationship of indoleacetic acid and tryptophan to dormancy and preharvest sprouting of wheat[J]. Functional Plant Biology, 2003, 30(9): 939-945. |
| [48] | Liu X, Zhang H, Zhao Y, Feng Z, Li Q, Yang H Q, Luan S, Li J, He Z H. Auxin controls seed dormancy through stimulation of abscisic acid signaling by inducing ARF-mediated ABI3 activation in Arabidopsis[J]. Proceedings of the National Academy of Sciences of the United States of America, 2013, 110(38): 15485-15490. |
| [49] | Mallory A C, Bartel D P, Bartel B. microRNA-directed regulation of Arabidopsis AUXIN RESPONSE FACTOR17 is essential for proper development and modulates expression of early auxin response genes[J]. The Plant Cell, 2005, 17(5): 1360-1375. |
| [50] | Wang J W, Wang L J, Mao Y B, Cai W J, Xue H W, Chen X Y. Control of root cap formation by microRNA-targeted auxin response factors in Arabidopsis[J]. The Plant Cell, 2005, 17(8): 2204-2216. |
| [51] | Chapman E J, Estelle M. Mechanism of auxin-regulated gene expression in plants[J]. Annual Review of Genetics, 2009, 43: 265-285. |
| [52] | Ogawa M, Hanada A, Yamauchi Y, Kuwahara A, Kamiya Y, Yamaguchi S. Gibberellin biosynthesis and response during Arabidopsis seed germination[J]. The Plant Cell, 2003, 15(7): 1591-1604. |
| [53] | Kurepa J, Li Y, Smalle J A. Cytokinin signaling stabilizes the response activator ARR1[J]. The Plant Journal, 2014, 78(1): 157-168. |
| [54] | Zhao Z, Andersen S U, Ljung K, Dolezal K, Miotk A, Schultheiss S J, Lohmann J U. Hormonal control of the shoot stem-cell niche[J]. Nature, 2010, 465(7301): 1089-1092. |
| [55] | Nie L, Song S, Yin Q, Zhao T, Liu H, He A, Wang W. Enhancement in seed priming-induced starch degradation of rice seed under chilling stress via GA-mediated α-amylase expression[J]. Rice, 2022, 15(1): 19. |
| [56] | Bailly C, El-Maarouf-Bouteau H, Corbineau F. From intracellular signaling networks to cell death: The dual role of reactive oxygen species in seed physiology[J]. Comptes Rendus Biologies, 2008, 331(10): 806-814. |
| [57] | El-Maarouf-Bouteau H, Bailly C. Oxidative signaling in seed germination and dormancy[J]. Plant Signaling & Behavior, 2008, 3(3): 175-182. |
| [58] | Foyer C H, Noctor G. Oxidant and antioxidant signalling in plants: A re-evaluation of the concept of oxidative stress in a physiological context[J]. Plant, Cell & Environment, 2005, 28(8): 1056-1071. |
| [59] | Gill S S, Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants[J]. Plant Physiology and Biochemistry, 2010, 48(12): 909-930. |
| [60] | Zavaliev R, Dong X. NPR1, a key immune regulator for plant survival under biotic and abiotic stresses[J]. Molecular Cell, 2024, 84(1): 131-141. |
| [61] | Gao W, Liu Y, Huang J, Chen Y, Chen C, Lu L, Zhao H, Men S, Zhang X. MES7 modulates seed germination via regulating salicylic acid content in Arabidopsis[J]. Plants, 2021, 10(5): 903. |
| [62] | Wani A B, Chadar H, Wani A H, Singh S, Upadhyay N. Salicylic acid to decrease plant stress[J]. Environmental Chemistry Letters, 2017, 15(1): 101-123. |
| [63] | Mishra P, Dubey R S. Effect of aluminium on metabolism of starch and sugars in growing rice seedlings[J]. Acta Physiologiae Plantarum, 2008, 30(3): 265-275. |
| [64] | Gómez-Cadenas A, Zentella R, Walker-Simmons M K, Ho T H. Gibberellin/abscisic acid antagonism in barley aleurone cells: Site of action of the protein kinase PKABA1 in relation to gibberellin signaling molecules[J]. The Plant Cell, 2001, 13(3): 667-679. |
| [65] | Wang E, Xu X, Zhang L, Zhang H, Lin L, Wang Q, Li Q, Ge S, Lu B R, Wang W, He Z. Duplication and independent selection of cell-wall invertase genes GIF1 and OsCIN1 during rice evolution and domestication[J]. BMC Evolutionary Biology, 2010, 10: 108. |
| [66] | He Y, Zhu M, Li Z, Jiang S, He Z, Xu S, Chen X, Hu Z, Zhang Z. IPA1 negatively regulates early rice seedling development by interfering with starch metabolism via the GA and WRKY pathways[J]. International Journal of Molecular Sciences, 2021, 22(12): 6605. |
| [67] | Ishibashi Y, Kasa S, Sakamoto M, Aoki N, Kai K, Yuasa T, Hanada A, Yamaguchi S, Iwaya-Inoue M. A role for reactive oxygen species produced by NADPH oxidases in the embryo and aleurone cells in barley seed germination[J]. PLoS One, 2015, 10(11): e0143173. |
| [1] | 黄奇娜, 姜鸿瑞, 杨婕, 于坤宇, 杨长登, 梁燕. 种子休眠基因Sdr4的生物信息学分析与分子标记开发和应用[J]. 中国水稻科学, 2026, 40(1): 61-71. |
| [2] | 郑广杰, 叶昌, 朱均林, 陶怡, 肖德顺, 徐亚楠, 褚光, 徐春梅, 王丹英. 淹水胁迫下水稻种子和胚芽葡萄糖供应差异与胚芽存活的关系[J]. 中国水稻科学, 2024, 38(2): 172-184. |
| [3] | 向镜, 陈惠哲, 张玉屏, 张义凯, 朱德峰. 淹涝条件下水温对水稻幼苗形态和生理的影响[J]. 中国水稻科学, 2016, 30(5): 525-531. |
| [4] | 马殿荣*,孔德秀,刘晓亮,高齐,丁国华,赵明辉,唐亮,徐正进,陈温福*. 杂草稻中胚轴伸长动态及其与籽粒淀粉酶活性和可溶性糖含量的关系[J]. 中国水稻科学, 2014, 28(1): 97-102. |
| [5] | 王娜1,任佐华1,邓林伟2,陈娟芳3 ,刘二明1,*. 稻曲病菌休眠与非休眠厚垣孢子胞壁多糖结构与组分[J]. 中国水稻科学, 2012, 26(3): 356-360. |
| [6] | 徐保钦, 陆作楣,. 杂交籼稻亲本种子休眠性与F1的相关及配合力研究 [J]. 中国水稻科学, 2008, 22(6): 583-583~589 . |
| [7] | 胡泽友邓小波,彭喜旭,何艳,刘文海,戴光宇,王海华,. 外源钙对镍胁迫下水稻幼苗抗氧化酶活性及膜脂过氧化的影响[J]. 中国水稻科学, 2007, 21(4): 367-371 . |
| [8] | 高东迎, 黄雪清, 刘永锋, 孙立华, 陈志谊, 刘蔼民. 水稻体细胞突变体HX-3在细胞水平上对白叶枯病的抗性[J]. 中国水稻科学, 2003, 17(4): 311-314 . |
| [9] | 陶龙兴, 王熹, 黄效林. 内源IAA对杂交稻强、弱势粒灌浆增重的影响[J]. 中国水稻科学, 2003, 17(2): 149-155 . |
| [10] | 朱 诚, 傅亚萍, 孙宗修. 超高产水稻开花结实期间叶片衰老与活性氧代谢的关系[J]. 中国水稻科学, 2002, 16(4): 326-330 . |
| [11] | 曾富华,吴岳轩,罗泽民. 生物及非生物诱导因子对水稻白叶枯病的诱导抗性及其与活性氧代谢的关系[J]. 中国水稻科学, 1999, 13(3): 165-169 . |
| [12] | 张春来,焦德茂,童红玉. 不同水稻品种的光氧化差异及活性氧清除剂的效应[J]. 中国水稻科学, 1993, 7(3): 175-178 . |
| [13] | 赵成章, 戚秀芳, 于飞. 休眠与水稻成熟胚细胞脱分化间的关系[J]. 中国水稻科学, 1989, 3(2): 73-76 . |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||