
中国水稻科学 ›› 2023, Vol. 37 ›› Issue (6): 563-576.DOI: 10.16819/j.1001-7216.2023.230301
收稿日期:2023-03-30
修回日期:2023-04-20
出版日期:2023-11-10
发布日期:2023-11-14
通讯作者:
*email: zhywang@zju.edu.cn
基金资助:
TONG Qi, WANG Chunyan, QUE Yawei, XIAO Yu, WANG Zhengyi*( )
)
Received:2023-03-30
Revised:2023-04-20
Online:2023-11-10
Published:2023-11-14
Contact:
*email: zhywang@zju.edu.cn
摘要:
【目的】 探明稻瘟病菌热激蛋白(HSP)40在形态分化和致病过程中的作用。【方法】 利用DNA同源重组方法敲除了稻瘟病菌HSP40编码基因MoMHF6,获得ΔMomhf6突变体,并通过表型分析、基因回补、RNA-seq分析等对MoMHF6的生物学功能进行较系统的研究。【结果】 敲除MoMHF6基因导致稻瘟病菌气生菌丝生长、无性产孢、孢子萌发、子囊壳产生和附着胞形成均显著下降,但与野生菌株相比,ΔMomhf6突变体在CM培养基上的径向生长没有显著差异,在子囊壳中仍可形成子囊和子囊孢子。在1、2和3 mol/L甘油溶液处理条件下,ΔMomhf6突变体附着胞塌陷率显著升高,说明MoMHF6与附着胞膨压形成有关。ΔMomhf6突变体对洋葱内表皮的穿透能力和对感病寄主的致病性完全丧失,甚至在划伤大麦叶片表皮组织中的扩展也受明显限制。而且,ΔMomhf6突变体对氧化胁迫的敏感性增强,附着胞发育过程中糖原的转运和降解缓慢,说明MoMHF6参与氧化胁迫反应和附着胞的糖原代谢。将完整的MoMHF6基因重新导入ΔMomhf6突变体可回补突变体的所有表型缺陷。另外,RNA-seq分析显示,部分已知的稻瘟病菌致病相关基因表达可能受MoMHF6调控,如MoATG4、MoPL1、MoVPR和GAS1等。【结论】 综上所述,稻瘟病菌HSP40编码基因MoMHF6在产孢、附着胞形成、穿透寄主、氧化胁迫应答、致病等过程中起重要作用,研究结果对进一步阐明MoMHF6调控稻瘟病菌形态分化和致病过程的基因网络和分子机制具有重要意义。
童琪, 王春燕, 阙亚伟, 肖宇, 王政逸. 稻瘟病菌热激蛋白(HSP)40编码基因MoMHF6的鉴定及功能研究[J]. 中国水稻科学, 2023, 37(6): 563-576.
TONG Qi, WANG Chunyan, QUE Yawei, XIAO Yu, WANG Zhengyi. Identification and Functional Characterization of the Heat Shock Protein (HSP) 40 Encoding Gene, MoMHF6, in Magnaporthe oryzae[J]. Chinese Journal OF Rice Science, 2023, 37(6): 563-576.
| 引物 Primer | 序列 Sequence (5′-3′) |
|---|---|
| MHF6-up-F | CCCCCGGGCTGCAGGAATTCGAGATAACAAAAGGTAT |
| MHF6-up-R | GCTCCTTCAATATCATCTTCTCTCGCTCAGTTCGAAATGGGAT |
| MHF6-down-F | TAGAGTAGATGCCGACCGAACAAGAATCAATGCCCAGTCTCGTGC |
| MHF6-down-R | TACCGGGCCCCCCCTCGAGCGAATACACTTTGGAGAC |
| HPT-F | GACAGACGTCGCGGTGAGTT |
| HPT-R | GTCCGAGGGCAAAGAAATAG |
| MHF6-YW-F | TCGAGGAGATTGAGTGCGTC |
| MHF6-YW-R | CTTCTTTTGCTTGGCTTTGC |
| MHF6-DX-F | GACTTCAATCAACCCTAACC |
| MHF6-DX-R | AAGCCGGACGGAAAGACTTT |
| HB-MHF6-F | TCCCCCGGGCTGCAGGAATTCTTCTCCAGAAAATCCCTGGA |
| HB-MHF6-R | GATAAGCTTGATATCGAATTCATTCGACCGGCGATCTTCCG |
| Actin-RT-F | ATTTACGAGGGTTTCTCCTTGC |
| Actin-RT-R | TCTCCTGCTCAAAGTCAAGAG |
| HOX2-qRT-F | CGATAATTGCTCCCACACCT |
| HOX2-qRT-R | GAAGGAGTCGGTGGTGACAT |
| COS1-qRT-F | ATGGATTCCCAGCCTCGTA |
| COS1-qRT-R | CGTTGACCAGCAAAGACAA |
| HTF1-qRT-F | GGCGACGATACGAAGAAA |
| HTF1-qRT-R | TGAACCACCTTGGCTTTG |
| CON7-qRT-F | GGCGACGATACGAAGAAA |
| CON7-qRT-R | TGAACCACCTTGGCTTTG |
| COM1-qRT-F | GAAAGAACCTATCAGGGCG |
| COM1-qRT-R | GTTTGCGATTGGCATTAGC |
| STU1-qRT-F | CTACGTTAAGTCCGAGATGG |
| STU1-qRT-R | CGTGATCAGCCTCATCTTCC |
表1 本研究中使用的引物
Table 1. Primers used in this study.
| 引物 Primer | 序列 Sequence (5′-3′) |
|---|---|
| MHF6-up-F | CCCCCGGGCTGCAGGAATTCGAGATAACAAAAGGTAT |
| MHF6-up-R | GCTCCTTCAATATCATCTTCTCTCGCTCAGTTCGAAATGGGAT |
| MHF6-down-F | TAGAGTAGATGCCGACCGAACAAGAATCAATGCCCAGTCTCGTGC |
| MHF6-down-R | TACCGGGCCCCCCCTCGAGCGAATACACTTTGGAGAC |
| HPT-F | GACAGACGTCGCGGTGAGTT |
| HPT-R | GTCCGAGGGCAAAGAAATAG |
| MHF6-YW-F | TCGAGGAGATTGAGTGCGTC |
| MHF6-YW-R | CTTCTTTTGCTTGGCTTTGC |
| MHF6-DX-F | GACTTCAATCAACCCTAACC |
| MHF6-DX-R | AAGCCGGACGGAAAGACTTT |
| HB-MHF6-F | TCCCCCGGGCTGCAGGAATTCTTCTCCAGAAAATCCCTGGA |
| HB-MHF6-R | GATAAGCTTGATATCGAATTCATTCGACCGGCGATCTTCCG |
| Actin-RT-F | ATTTACGAGGGTTTCTCCTTGC |
| Actin-RT-R | TCTCCTGCTCAAAGTCAAGAG |
| HOX2-qRT-F | CGATAATTGCTCCCACACCT |
| HOX2-qRT-R | GAAGGAGTCGGTGGTGACAT |
| COS1-qRT-F | ATGGATTCCCAGCCTCGTA |
| COS1-qRT-R | CGTTGACCAGCAAAGACAA |
| HTF1-qRT-F | GGCGACGATACGAAGAAA |
| HTF1-qRT-R | TGAACCACCTTGGCTTTG |
| CON7-qRT-F | GGCGACGATACGAAGAAA |
| CON7-qRT-R | TGAACCACCTTGGCTTTG |
| COM1-qRT-F | GAAAGAACCTATCAGGGCG |
| COM1-qRT-R | GTTTGCGATTGGCATTAGC |
| STU1-qRT-F | CTACGTTAAGTCCGAGATGG |
| STU1-qRT-R | CGTGATCAGCCTCATCTTCC |
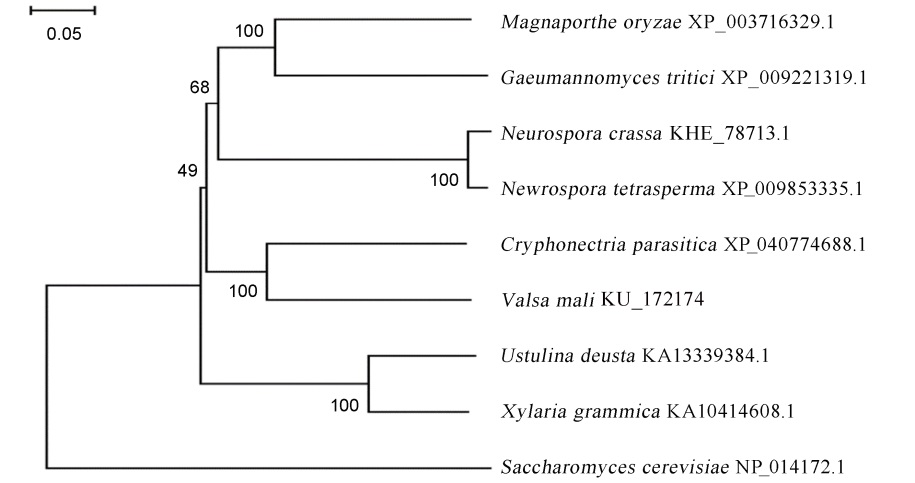
图1 MoMhf6与部分真菌同源蛋白的系统发育分析 通过MEGA 7.0 软件对包括稻瘟病菌在内的9个真菌的HSP40同源蛋白构建系统进化树。进化树节点上的数字代表1000株树中有百分之几的树具有这根树枝,即自展值,反映了该树枝的可信度。树枝的长短代表遗传距离。进化树的距离标尺为0.05。
Fig. 1. Phylogenetic analysis of MoMhf6 and its orthologues from several fungal species. The phylogenetic tree was constructed by MEGA version 7.0. Numbers at nodes of the branch represent bootstrapping value in 1000 replications. The length of the branches represents genetic distance. The distance scale=0.05.
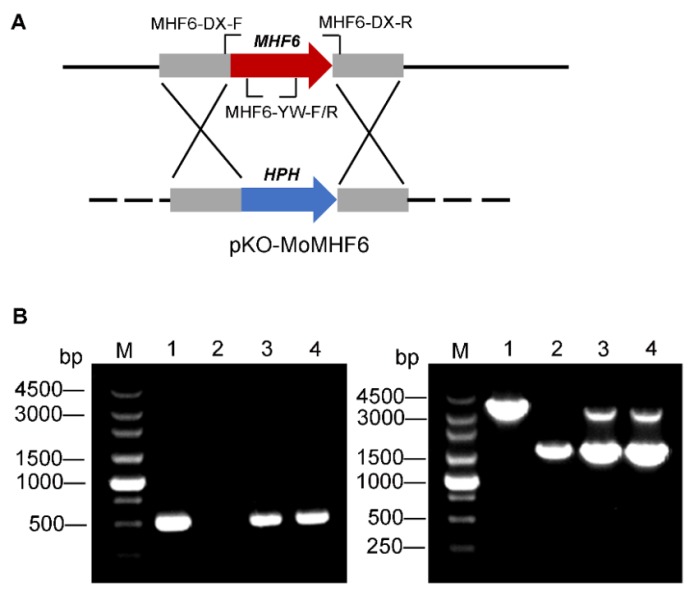
图2 MoMHF6基因敲除示意图及ΔMomhf6突变体和互补转化子的PCR验证 A―敲除载体pKO-MoMHF6及MoMHF6靶标基因的敲除,红箭头表示靶基因MoMHF6,蓝箭头代表耐潮霉素基因,图中引物位置被标出;B―确认缺失突变体ΔMomhf6和回补菌株ΔMomhf6-C,左图:用MHF6-YW-F/R引物扩增出524 bp的MoMHF6基因片段;右图:用MHF6-DX-F/R引物验证不同菌株靶向基因缺失或回补,1.5 kb和3.14 kb的条带分别对应HPH和MoMHF6。M−Marker; 泳道1~4分别代表Guy11, ΔMomhf6, MoMHF6-ect(ectopic)和ΔMomhf6-C (complemented)。
Fig. 2. Schematic diagram of gene knockout of MoMHF6 and confirmation of ΔMomhf6 mutants and complemented transformants by PCR. A, Knockout vector pKO-MoMHF6 and target gene deletion of MoMHF6. The red arrow represents the target gene MoMHF6. The blue arrow represents the hygromycin resistant gene. The location of primers is indicated; B, Confirmation of the deletion mutant ΔMomhf6 and the complemented strain ΔMomhf6-C. Left: A 524 bp MoMHF6 gene fragment was amplified by primers MHF6-YW-F/R. Right: the targeted gene deletion or complementation events of different strains were further verified by primers MHF6-DX-F/R. The bands of about 1.5 kb and 3.14 kb were corresponding with HPH and MoMHF6 genes respectively. M, Marker; Lane 1, Guy11; Lane 2, ΔMomhf6; Lane 3, MoMHF6-ect (ectopic); Lane 4, ΔMomhf6-C (complemented).
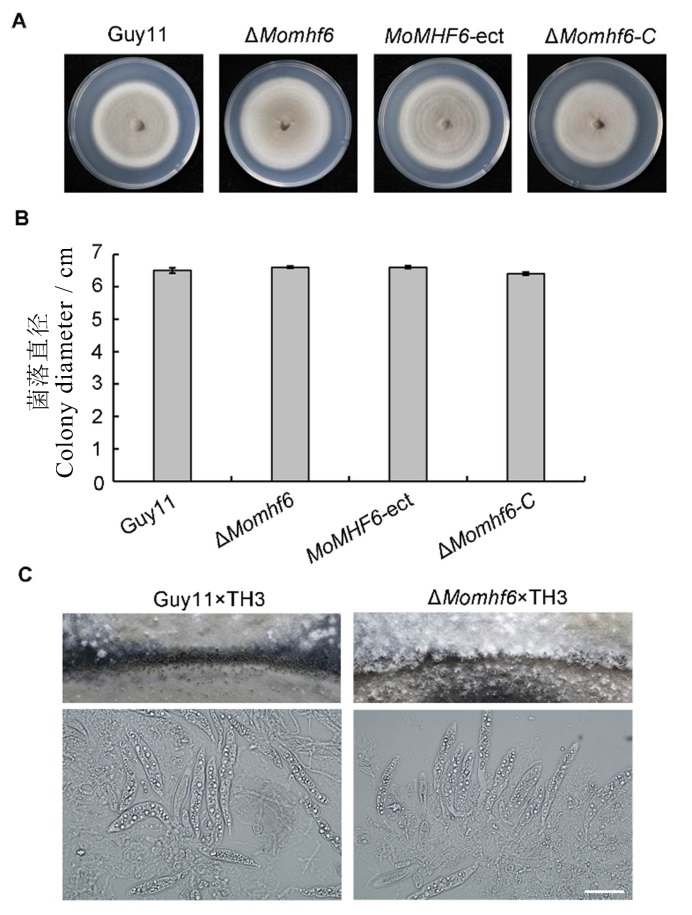
图3 MoMHF6是气生菌丝生长和子囊壳产生所必需的 A−将野生型Guy11、ΔMomhf6突变体、异位插入菌株MoMHF6-ect和回补菌株ΔMomhf6-C在CM板上25℃下培养10 d,接种10 d后拍照;B−柱状图代表不同菌株在CM培养基上的径向生长,误差线表示标准误;C−ΔMomhf6×TH3杂交组合的子囊壳显著减少,在两种杂交组合中均能观察到子囊和子囊孢子,比例尺为20 μm。
Fig. 3. MoMHF6 is required for aerial hypha growth and perithecia production. A, The wild type Guy11, the ΔMomhf6 mutant, the ectopic insertion strain MoMHF6-ect and the complemented strain ΔMomhf6-C were cultured on CM plates at 25 ℃ for 10 days and photographed at 10 days after inoculation; B, The bar graph shows radical growth of different strains on CM plates. Error bars represent standard error; C, Perithecia production was severely reduced for the ΔMomhf6×TH3 cross. Asci and ascospores in perithecia of both crosses were observed. Bar=20 μm.
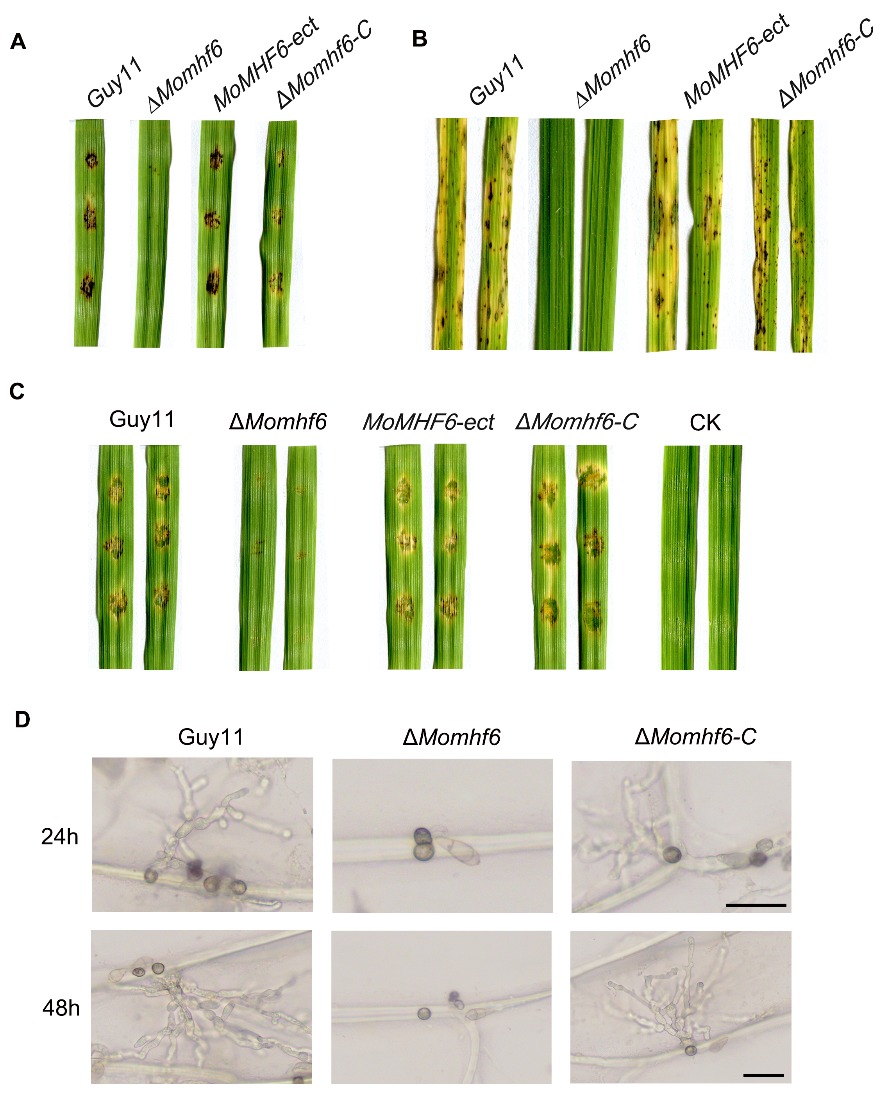
图4 MoMHF6在稻瘟病菌致病过程中具有重要作用 A―ΔMomhf6突变体对易感病的大麦叶片无致病性。将不同菌株的分生孢子悬液(浓度为5×104 个/mL)点接于大麦叶段,接种5 d后拍照;B―ΔMomhf6突变体对易感病的水稻叶片无致病性。将不同菌株的分生孢子悬液(浓度为5×104 个/mL)喷雾接种于水稻幼苗上,接种5 d后拍照;C―ΔMomhf6突变体在划伤大麦叶片上的侵染生长受到明显抑制。不同菌株的分生孢子悬液(浓度为5×104个/mL)点接于划伤的大麦叶段上,CK表示H2O,接种5 d后拍照;D―ΔMomhf6突变体不能穿透洋葱表皮质层和细胞壁。将不同菌株的分生孢子悬液(浓度为2×104 个/mL)点接于洋葱表皮上,在黑暗条件下诱导24 h和48 h后,在光学显微镜下进行镜检和拍照,比例尺为20 μm。
Fig. 4. MoMHF6 plays important roles in pathogenesis by M. oryzae. A, The ΔMomhf6 mutant is nonpathogenic to susceptible barley leaves. Conidial suspension (5×104 conidia/mL) of different tested strains were drop-inoculated onto barley leaf segments and photographed at 5 dpi (days post inoculation); B, The ΔMomhf6 mutant is nonpathogenic to susceptible rice leaves. Rice seedlings were sprayed with conidial suspension (5×104 conidia/mL) of different strains and photographed at 5 dpi; C, Invasive growth of the ΔMomhf6 mutant was severely impaired on wounded barley leaf segments. Conidial suspension (5×104 conidia/mL) of different strains was dropped on wounded barley leaf segments. CK represents H2O. Photographs were taken at 5 dpi; D, The ΔMomhf6 mutant was unable to penetrate onion cuticle and cell wall. Conidial suspension (2×104 conidia/mL) of different strains was dropped on onion epidermis. After 24 h and 48 h of incubation in darkness, the onion epidermis was examined and photographed under a light microscope. Bars=20 μm.
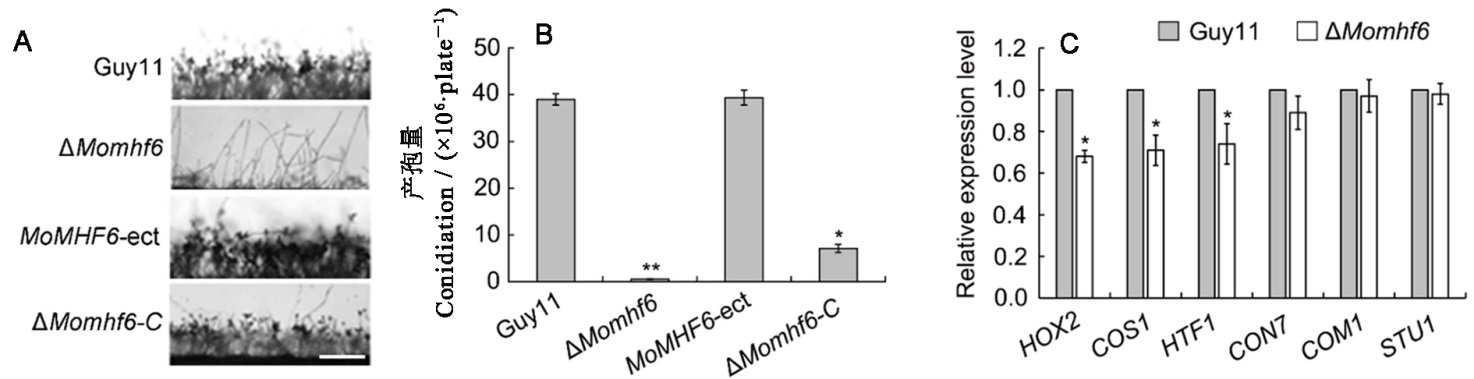
图5 MoMHF6在稻瘟病菌无性产孢中起重要作用 A―显微镜下观察气生菌丝生长和分生孢子梗发育,比例尺为100 μm;B―25℃条件下,不同菌株在CM培养基上培养12 d后统计分析产孢量;C―qRT-PCR分析几个产孢相关基因转录表达水平,误差线表示标准误,星号表示显著差异(*P<0.05; **P<0.01)。
Fig. 5. MoMHF6 is important for asexual sporulation. A, Microscopic observation of aerial hypha growth and conidiophore development. Bars=100 μm; B, Statistical analysis of conidiation on CM at 25 ℃ for 12 days; C, qRT-PCR analysis of transcriptional expression of several sporulation related genes. Error bars represent standard error. Asterisks indicate significant difference (*P<0.05; **P<0.01).
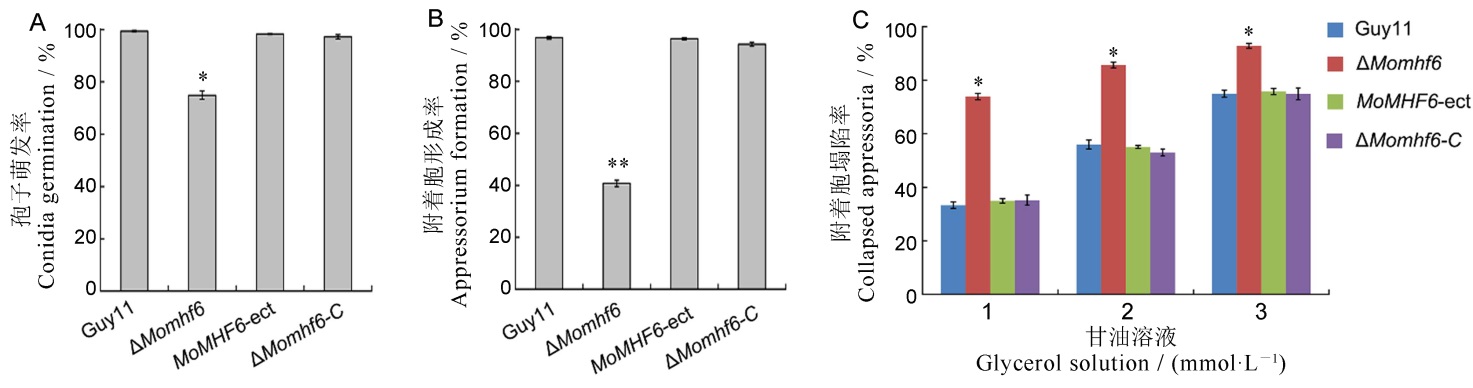
图6 敲除MoMHF6导致分生孢子萌发、附着胞形成和附着胞膨压积累显著降低 A―统计分析分生孢子萌发率,分生孢子在疏水表面诱导萌发6 h;B―统计分析附着胞形成率,将分生孢子置于疏水表面诱导形成附着胞,黑暗诱导24 h;C―统计分析附着胞塌陷率,用1 mol/L、2 mol/L、3 mol/L的甘油溶液处理诱导24 h形成的附着胞,处理时间10 min,误差线表示标准误,星号表示显著差异(*P<0.05; **P<0.01)。
Fig. 6. Deletion of MoMHF6 results in significant reduction of conidium germination, appressorium formation and appressorium turgor generation A, Statistical analysis of conidium germination. Conidia were allowed to germinate on hydrophobic surface for 6 hours; B, Statistical analysis of appressorium formation. Conidia were placed on the hydrophobic surface to induce appressorium formation and incubated for 24 hours; C, Statistical analysis of collapsed appressoria. Appressoria induced for 24 hours were incubated in 1 mol/L, 2 mol/L or 3 mol/L glycerol solution for 10 minutes. Error bars represent standard error. Asterisks indicate significant difference (*P<0.05; **P<0.01).
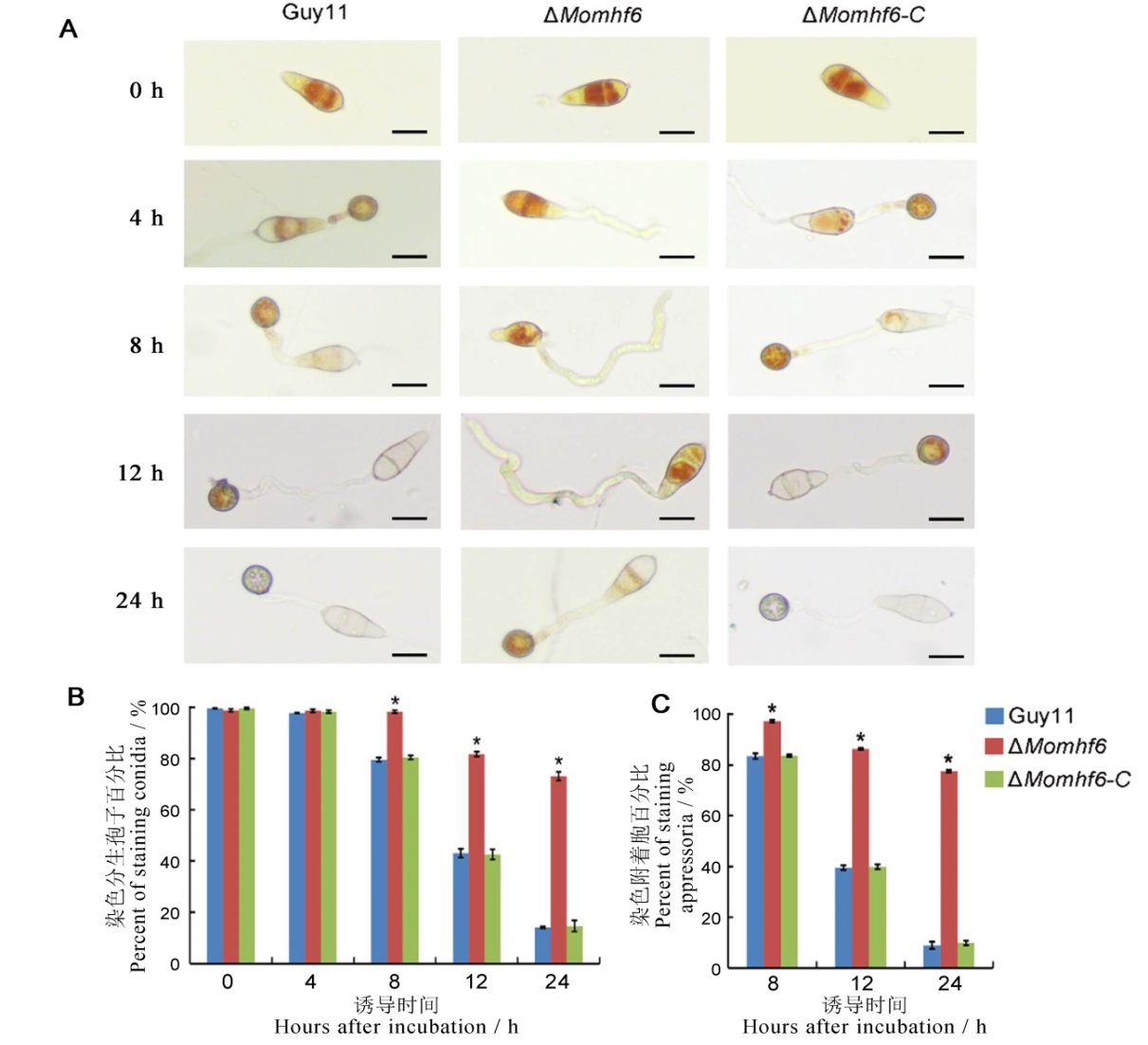
图7 MoMHF6基因的缺失导致稻瘟病菌附着胞发育过程中糖原转运和降解延缓 A-将野生型Guy11、ΔMomhf6和回补菌株ΔMomhf6-C的分生孢子悬液(浓度为5×104 个/mL)分别在疏水表面诱导4、8、12、24 h后,用KI/I2溶液进行染色,并在不同的时间间隔拍照,比例尺为20 μm;B-统计各待测菌株在不同时间段分生孢子内的糖原转移情况;C-统计各待测菌株在不同时间段附着胞内的糖原降解情况。误差线表示标准误,星号表示显著差异(P<0.05)。
Fig. 7. Deletion of MoMHF6 delays glycogen transportation and degradation during appressoria development in M. oryzae. A, Conidial suspension (5×104 conidia/mL) of the wild type Guy11, the mutant ΔMomhf6 and the complemented strain ΔMomhf6-C was incubated on hydrophobic surface for 4, 8, 12, 24 hours respectively and then stained with KI/I2 solution. Photographs were taken at different time intervals. Bars=20 μm. B, Statistical analysis of glycogen mobilization in conidia of different tested strains; C, Statistical analysis of glycogen degradation in appressoria of different tested strains. Error bars represent standard error. Asterisks indicate significant difference (P<0.05).
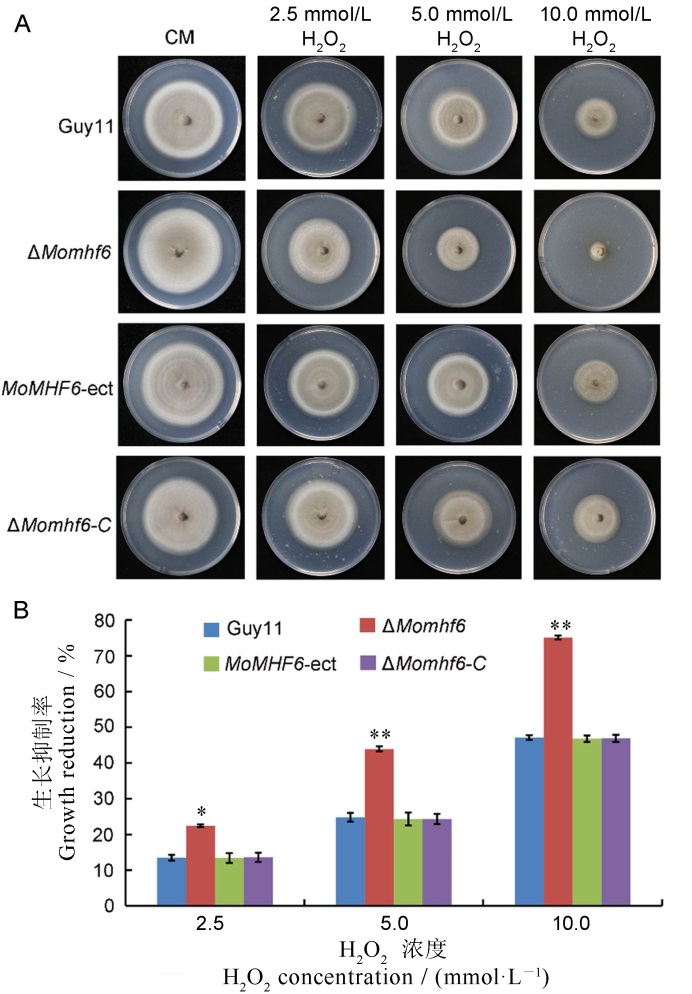
图8 MoMHF6缺失增强稻瘟病菌对氧化胁迫的敏感性 A-将不同菌株分别接种在含有2.5、5和10 mmol/L H2O2的CM培养基上,培养10 d后观察菌落形态;B-测定菌落直径并计算生长抑制率,误差线表示标准误,星号表示显著差异 (*P<0.05; **P < 0.01)。
Fig. 8. Deletion of MoMHF6 increases sensitivity of M. oryzae to oxidative stress. A, The different tested strains were inoculated on CM supplemented with 2.5, 5 and 10 mmol/L H2O2, respectively. Growth of various tested strains after 10 days is presented; B, Colony diameters were measured and growth inhibition rates were calculated. Error bars represent standard deviation. Asterisks indicate significant difference (*P<0.05; **P<0.01).
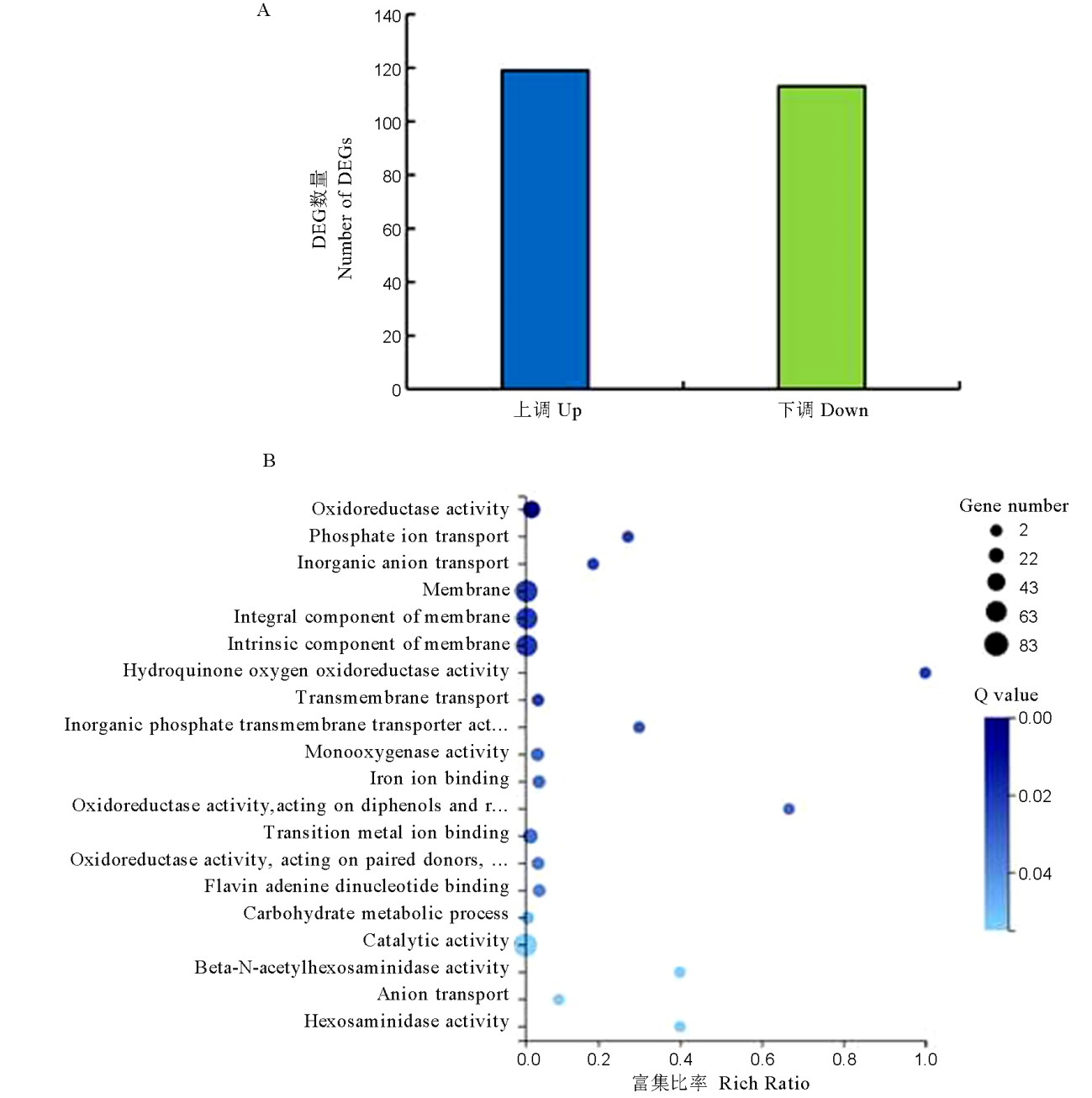
图9 ΔMomhf6突变体转录组差异表达基因分析 A—ΔMomhf6差异表达基因数量的统计分析;B—ΔMomhf6突变体DEGs的GO富集散点图,富集比率表示GO项中标注的DEGs数量与同一GO项中标注的所有基因数量的比值,q值是修正后的p值,q值越小表示集中度越高。
Fig. 9. Transcriptome analysis of the ΔMomhf6 mutant for differentially expressed genes. A, Statistical analysis of numbers of differentially expressed genes in the ΔMomhf6 mutant in comparison to the wild type strain Guy11; B, Scatter plot of GO term enrichment of DEGs in the ΔMomhf6 mutant. Rich ratio represents the ratio of numbers of DEGs annotated in the GO term to the numbers of all genes annotated in the same GO term. The q-value is corrected p value, with lower value means greater intensiveness.
| [1] | Talbot N J. On the trail of a cereal killer: Exploring the biology of Magnaporthe grisea[J]. Annual Review of Microbiology, 2003, 57(1): 177-202. |
| [2] | Muthayys S, Sugimoto J D, Montgomery S, Maberly G F. An overview of global rice production, supply, trade, and consumption[J]. Annals of the New York Academy of Sciences, 2015, 1324(1): 7-14. |
| [3] | 商文奇. 水稻稻瘟病防治方法研究进展[J]. 辽宁农业科学, 2021(1): 33-39. |
| Shang W Q. Advances in research on control methods of rice blast[J]. Liaoning Agricultural Science, 2021(1): 33-39. (in Chinese) | |
| [4] | Cyr D M, Lu X, Douglas M G. Regulation of Hsp70 function by a eukaryotic DnaJ homolog[J]. Journal of Biological Chemistry, 1992, 267: 20927-20931. |
| [5] | Hennessy F, Nicoll W S, Zimmermann R, Cheetham M E,; Blatch G L. Not all J domains are created equal: Implications for the specificity of Hsp40-Hsp70 interactions[J]. Protein Science, 2005, 14(7): 1697-1709. |
| [6] | Szyperski T, Pellecchia M, Wall D, Georgopoulos C, Wuthrich K. NMR structure determination of the Escherichia coli DnaJ molecular chaperone: Secondary structure and backbone fold of the N-terminal region (residues 2-108) containing the highly conserved J domain[J]. Proceedings of the National Academy of Sciences of the United States of America, 1994, 91(24): 11343-11347. |
| [7] | Tamadaddi C A, Sahi C. J domain independent functions of J proteins[J]. Cell Stress Chaperones, 2016, 21(4): 563-570. |
| [8] | Kelley W L. The J-domain family and the recruitment of chaperone power[J]. Trends in Biochemical Sciences, 1998, 23(6): 222-227. |
| [9] | Liberek K, Lewandowska A, Zietkiewicz S. Chaperones in control of protein disaggregation[J]. EMBO Journal, 2008, 27(2): 328-335. |
| [10] | Meyer A E, Hung N J, Yang P, Johnson A W, Craig E A. The specialized cytosolic J-protein, Jjj1, functions in 60S ribosomal subunit biogenesis[J]. Proceedings of the National Academy of Sciences of the United States of America, 2007, 104(5): 1558-1563. |
| [11] | Lim J G, Lee J G, Kim J M. A DnaJ-like homolog from Cryphonectria parasitica is not responsive to hypoviral infection but is important for fungal growth in both wild-type and hypovirulent strains[J]. Molecules & Cells, 2010, 30(3): 235-243. |
| [12] | Yi M H, Lee Y H. Identification of genes encoding heat shock protein 40 family and the functional characterization of two Hsp40s, MHF16 and MHF21, in Magnaporthe oryzae[J]. The Plant Pathology Journal, 2008, 24(2): 7271-7279. |
| [13] | Yang J, Liu M X, Liu X Y, Yin Z Y, Sun Y, Zhang H F, Zheng X B, Wang P, Zhang Z G. Heat-shock proteins MoSsb1, MoSsz1, and MoZuo1 attenuate MoMkk1-mediated cell-wall integrity signaling and are important for growth and pathogenicity of Magnaporthe oryzae[J]. Molecular Plant-Microbe Interaction, 2018, 31(11): 1211-1221. |
| [14] | Wang Z Y, Jenkinson J M, Holcombe L J. The molecular biology of appressorium turgor generation by the rice blast fungus Magnaporthe grisea[J]. Biochemical Society Transactions, 2005, 33(2): 384-388. |
| [15] | Badaruddin M, Holcombe L J, Wilson R A, Wang Z Y, Kershaw M J, Talbot N J. Glycogen metabolic genes are involved in trehalose-6-phosphate synthase-mediated regulation of pathogenicity by the rice blast fungus Magnaporthe oryzae[J]. PLoS Pathogens, 2013, 9(10): e1003604. |
| [16] | Thins E, Weber R W, Talbot N J. MAP kinase and protein kinase A-dependent mobilization of triacylglycerol and glycogen during appressorium turgor generation by Magnaporthe grisea[J]. Plant Cell, 2000, 12(9): 1703-1718. |
| [17] | Guo M, Chen Y, Du Y, Dong Y, Guo W, Zhai S, Zhang H, Dong S, Zhang Z, Wang Y, Wang P, Zheng X. The bZIP transcription factor MoAP1 mediates the oxidative stress response and is critical for pathogenicity of the rice blast fungus Magnaporthe oryzae[J]. PLoS Pathogens, 2011, 7(2): e1001302. |
| [18] | Lin C H, Yang S L, Chung K R. The YAP1 homolog-mediated oxidative stress tolerance is crucial for pathogenicity of the necrotrophic fungus Alternaria alternata in citrus[J]. Molecular Plant-Microbe Interactions, 2009, 22(8): 942-952. |
| [19] | Molina L, Kahmann R. An Ustilago maydis gene involved in H2O2 detoxification is required for virulence[J]. Plant Cell, 2007, 19(7): 2293-2309. |
| [20] | Liu T B, Liu X H, Lu J P, Zhang L, Min H, Lin F C. The cysteine protease MoAtg4 interacts with MoAtg8 and is required for differentiation and pathogenesis in Magnaporthe oryzae[J]. Autophagy, 2010, 6(1): 74-85. |
| [21] | Wegner A, Casanova F, Loehrer M, Jordine A, Bohnert S, Liu X Y, Zhang Z G, Schaffrath U. Gene deletion and constitutive expression of the pectate lyase gene 1 (MoPL1) lead to diminished virulence of Magnaporthe oryzae [J]. Journal of Microbiology, 2022, 60(1): 79-88. |
| [22] | Nguyen Q B, Itoh K, Van V B, Tosa Y, Nakayashiki H. Simultaneous silencing of endo-β-1,4 xylanase genes reveals their roles in the virulence of Magnaporthe oryzae[J]. Molecular Microbiology, 2011, 81(4): 1008-1019. |
| [23] | Qian B, Su X, Ye Z, Liu X Y, Liu M X, Shen D Y, Chen H, ZhangH F, Wang P, Zhang Z G. MoErv29 promotes apoplastic effector secretion contributing to virulence of the rice blast fungus Magnaporthe oryzae[J]. New Phytologist, 2021, 233(3): 1289-1302. |
| [24] | Oh Y, Donofrio N, Pan H, Coughlan S, Dean R A. Transcriptome analysis reveals new insight into appressorium formation and function in the rice blast fungus Magnaporthe oryzae[J]. Genome Biology, 2008, 9(5): R85. |
| [25] | Xue C Y, Park G, Choi W B. Two novel fungal virulence genes specifically expressed in appressoria of the rice blast fungus[J]. Plant Cell, 2002, 14(9): 2107-2119. |
| [26] | Geoghegan I A, Gurr S J. Investigating chitin deacetylation and chitosan hydrolysis during vegetative growth in Magnaporthe oryzae[J]. Cell Microbiology, 2017, 19(9): e12743. |
| [27] | 邹逸宾. 不同剂型1, 2-苯并异噻唑啉-3-酮对五种镰孢菌的效果评价和稻瘟病菌中MoAop1的功能分析[D]. 南京: 南京农业大学, 2019. |
| Zou Y B. Evaluation of different forms of BIT to five species of Fusarium and functional characterization of MoAop1 in Magnaporthe grisea[D]. Nanjing: Nanjing Agricultural University, 2019. (in Chinese with English abstract) | |
| [28] | Zhang H F, Liu K Y, Xing Z, Song W, Zhao Q, Dong Y, Guo M, Zheng X, Zhang Z. A two-component histidine kinase, MoSLN1, is required for cell wall integrity and pathogenicity of the rice blast fungus, Magnaporthe oryzae[J]. Current Genetics, 2010, 56(6): 517-528. |
| [29] | Bekh-Ochir D, Shimada S, Yamagami A, Kanda S, Ogawa K, Nakazawa M, Matsui M, Sakuta M, Osada H, Asami T, Nakano T. A novel mitochondrial DnaJ/HSP40 family protein BIL2 promotes plant growth and resistance against environmental stress in brassinosteroid signaling[J]. Planta, 2013, 237(6): 1509-1525. |
| [30] | Liu J Z, Whitham S A. Overexpression of a soybean nuclear localized type-III DnaJ domain-containing HSP40 reveals its roles in cell death and disease resistance[J]. Plant Journal, 2013, 74(1): 110-121. |
| [31] | Zhong X, Yang J, Shi Y. The DnaJ protein OsDjA6 negatively regulates rice innate immunity to the blast fungus Magnaporthe oryzae[J]. Molecular Plant Pathology, 2018, 19(3): 607-614. |
| [32] | Saunders D G, Dagdas Y F, Talbot N J. Spatial uncoupling of mitosis and cytokinesis during appressorium-mediated plant infection by the rice blast fungus Magnaporthe oryzae[J]. Plant Cell, 2010, 22(7): 2417-2428. |
| [33] | Fernandez J, Orth K. Rise of a cereal killer: The biology of Magnaporthe oryzae biotrophic growth[J]. Trends in Microbiology, 2018, 26(7): 582-597. |
| [34] | Howard R J, Ferrari M A, Roach D H. Penetration of hard substrates by a fungus employing enormous turgor pressures[J]. Proceedings of the National Academy of Sciences of the United States of America, 1991, 88(24): 11281-11284. |
| [35] | Wilson R A, Talbot N J. Under pressure: investigating the biology of plant infection by Magnaporthe oryzae[J]. Nature Reviews Microbiology, 2009, 7: 185-195. |
| [36] | Nishimura M, Fukada J, Moriwaki A, Fujikawa T, Hayashi N. Mstu1, an APSES transcription factor, is required for appressorium-mediated infection in Magnaporthe grisea[J]. Bioscience Biotechnology and Biochemistry, 2009, 73(8): 1779-1786. |
| [37] | Liu C, Shen N, Zhang Q. Magnaporthe oryzae transcription factor MoBZIP3 regulates appressorium turgor pressure formation during pathogenesis[J]. International Journal of Molecular Sciences, 2022, 23(2): 881. |
| [38] | Qiu X B, Shao Y M, Miao S, Wang L. The diversity of the DnaJ/Hsp40 family, the crucial partners for Hsp70 chaperones[J]. Cellular and Molecular Life Sciences, 2006, 63(22): 2560-2570. |
| [39] | Yan W, Schilke B, Pfund C, Walter W, Craig E A. Zuotin, a ribosome-associated DnaJ molecular chaperone[J]. EMBO Journal, 1998, 17(16): 4809-4817. |
| [1] | 刘树芳, 董丽英, 李迅东, 周伍民, 杨勤忠. 持有Pi9基因的水稻单基因系IRBL9-W对稻瘟病菌苗期和成株期抗性差异[J]. 中国水稻科学, 2021, 35(3): 303-310. |
| [2] | 孟峰, 张亚玲, 靳学慧. 黑龙江省稻瘟病菌无毒基因AVR-Pita及其同源基因的检测与分析[J]. 中国水稻科学, 2020, 34(2): 143-149. |
| [3] | 冯向阳, 张震, 柴荣耀, 邱海萍, 王教瑜, 毛雪琴, 王艳丽, 孙国昌. ATP硫酸化酶基因MoMET3在稻瘟病菌生长发育和致病过程中的功能分析[J]. 中国水稻科学, 2017, 31(5): 542-550. |
| [4] | 朱名海, 皮磊, 舒灿伟, 周而勋. 南繁区稻瘟病菌遗传多样性和群体遗传结构的AFLP分析[J]. 中国水稻科学, 2017, 31(3): 320-326. |
| [5] | 阮宏椿, 石妞妞, 杜宜新, 甘林, 杨秀娟, 代玉立, 陈福如. 水稻抗性基因Pi对福建省稻瘟病菌优势菌群的抗性分析[J]. 中国水稻科学, 2017, 31(1): 105-110. |
| [6] | 顾卓侃, 李玲, 王教瑜, 柴荣耀, 王艳丽, 张震, 毛雪琴, 邱海萍, 孙国昌. 利用卡氏白和尼罗红染色观察稻瘟病菌有性世代的结构[J]. 中国水稻科学, 2016, 30(6): 668-672. |
| [7] | 刘鑫, 张恒, 阚虎飞, 周立帅, 黄昊, 宋林林, 翟焕趁, 张君, 鲁国东. 水稻泛素结合酶基因家族的生物信息学与表达分析[J]. 中国水稻科学, 2016, 30(3): 223-231. |
| [8] | 王玲, 左示敏, 张亚芳, 陈宗祥, 潘学彪, 黄世文. 四川省稻瘟病菌群体遗传结构分析[J]. 中国水稻科学, 2015, 29(3): 327-334. |
| [9] | 李贺, 韩艺娟, 林艺娟, 刘丽华, 张承康, 张连虎, 王宗华, 鲁国东. 水稻锌指蛋白基因OsZFP1的功能分析[J]. 中国水稻科学, 2015, 29(2): 135-140. |
| [10] | 陈亚平, 施文骁, 王洪凯. 稻瘟病菌大片段DNA转化载体的构建[J]. 中国水稻科学, 2015, 29(1): 91-96. |
| [11] | 毛雪琴,姜华,王艳丽,张震,柴荣耀,王教瑜,邱海萍,杜新法,孙国昌*. 稻瘟病菌不同拼接版本基因组序列差异性比较[J]. 中国水稻科学, 2013, 27(4): 425-433. |
| [12] | 刘永锋, 陈志谊, 俞文渊, 于俊杰, 胡建坤, 刘邮洲, 聂亚锋. 稻瘟病菌分泌蛋白质在致病过程中的作用初探[J]. 中国水稻科学, 2011, 25(4): 452-454. |
| [13] | 王小艳,王教瑜,姜 华 ,王艳丽,张 震,柴荣耀,毛雪琴,邱海萍,杜新法,孙国昌,. 稻瘟病菌对新型模式植物二穗短柄草的致病性研究[J]. 中国水稻科学, 2011, 25(3): 314-320 . |
| [14] | 吴小燕 王教瑜张 震,井金学,杜新法,柴荣耀,毛雪琴,邱海萍, 姜 华,王艳丽,孙国昌,. 稻瘟病菌致病性增强突变体B11的基因分析[J]. 中国水稻科学, 2009, 23(6): 611-615 . |
| [15] | 高方远,陆贤军,何树林,陈晓娟,卢代华,孙淑霞,李治华,刘光春,张义正,任光俊. 转基因抗虫性水稻恢复系选育及特性分析[J]. 中国水稻科学, 2008, 22(4): 353-358 . |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||